Shenzhen Lawyer Attends FDA Regulatory Conference and Comments on Compliance Relief for Vaping Compa
On August 22, 2023, from 10 PM to 3:30 AM Beijing time on August 23, the U.S. FDA's Center for Tobacco Products (CTP) held an online hearing regarding the formulation of a five-year strategic plan. The meeting lasted for five and a half hours, during which Dr. Yang Aoyu from the Zheng Mingwei lawyer team of Zhongyin (Shenzhen) Law Firm participated throughout and published comments in the Federal Register.
1. Strategic Goals of the FDA's Center for Tobacco Products
The FDA emphasized five preliminary strategic goals at the meeting:
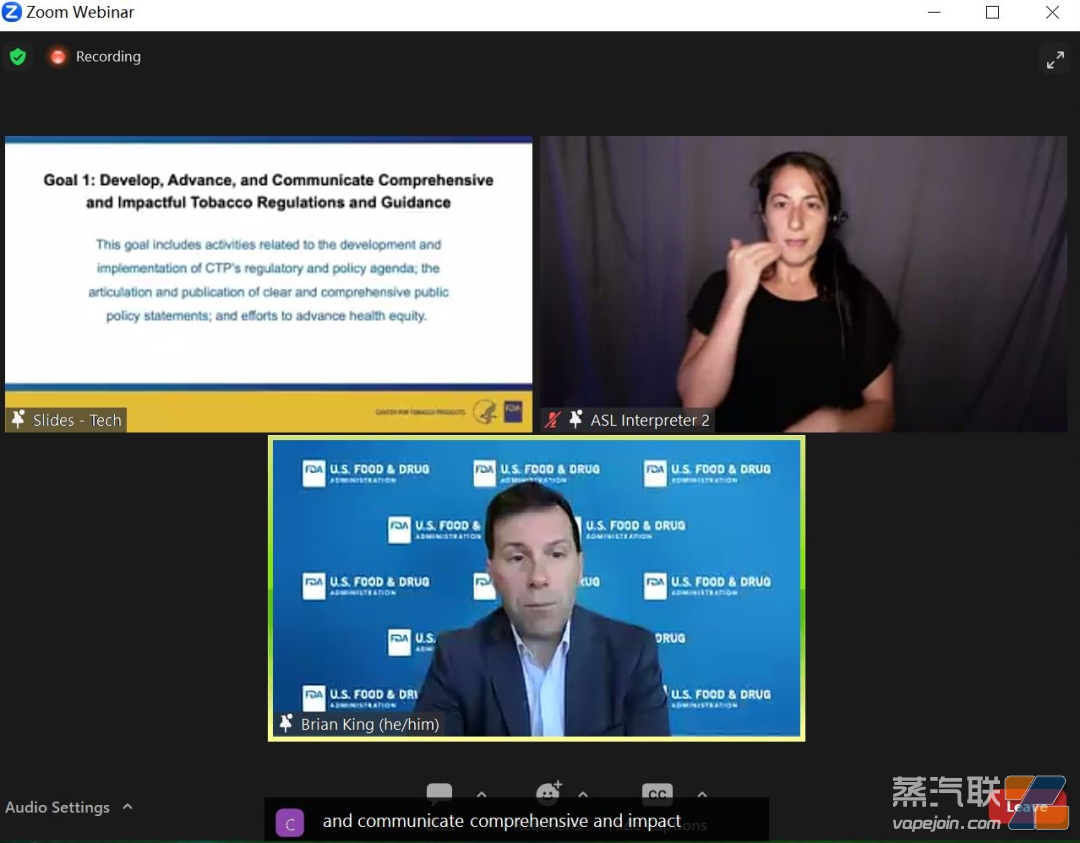
1. Develop, promote, and disseminate comprehensive and impactful tobacco regulations and guidelines.
2. Ensure timely, clear, and consistent product application reviews to protect public health.
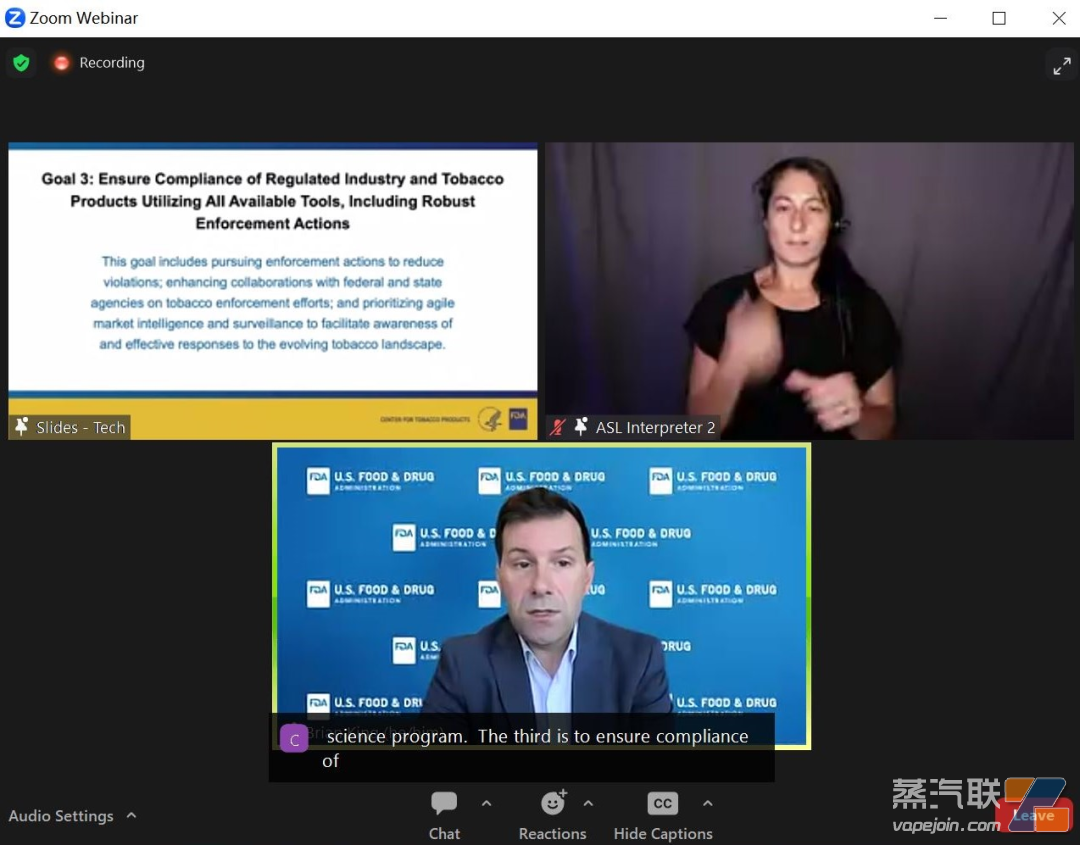
3. Utilize all available tools, including strong enforcement actions, to ensure compliance in the regulated industry and tobacco products.
4. Improve public health by increasing awareness and understanding of CTP tobacco product regulations and the risks associated with tobacco product use.
5. Advance operational excellence.
2. Public Opinions
Regarding the above strategic plan, various sectors of American society expressed opinions on the legal regulation of tobacco products. The opinions mainly focused on whether the sale of flavored e-cigarettes and menthol-flavored e-cigarettes should be banned, and some comments also questioned the transparency of the PMTA process and warning letter enforcement procedures. The meeting did not specifically address Chinese companies or Chinese e-cigarette brands, with only about three mentions that most products come from China.
1. Should the Sale of Flavored E-Cigarettes and Menthol-Flavored E-Cigarettes Be Banned?
Regarding whether the sale of flavored e-cigarettes and menthol-flavored e-cigarettes should be banned, most public welfare organizations, universities, and social research institutions support the ban, arguing that flavored e-cigarettes and menthol-flavored e-cigarettes do not effectively serve as substitutes for traditional cigarettes and instead attract youth, increasing non-smokers' use of tobacco products, thereby harming public health.
In contrast, small wholesalers, retailers, police, and the Black community largely oppose the ban on flavored e-cigarettes and menthol-flavored e-cigarettes. Small wholesalers and retailers argue that they primarily rely on selling tobacco products for their livelihoods, and the employment numbers and economic share of this group cannot be ignored. Therefore, a complete ban on flavored e-cigarettes and menthol-flavored e-cigarettes is unreasonable. If the FDA wants to reduce youth use of e-cigarettes, the dissemination of laws and regulations, student education, and parental supervision are more important. The police and the Black community's viewpoint is that banning flavored e-cigarettes and menthol-flavored e-cigarettes will lead to a significant increase in underground circulation of such e-cigarettes, increasing the enforcement workload; and due to unique racial issues in the U.S., the initial phase of the ban will inevitably target the Black community first, whether they are users or sellers.
2. Transparency of the PMTA Process
Some social groups, especially small manufacturers and retailers, believe that the PMTA application process is too complicated and the review process is opaque. The legal regulations regarding the documents that need to be submitted are difficult for ordinary people to understand, which is unfriendly to them. If they want to submit applications without relying on professionals like lawyers, it is almost impossible to complete the application process. Additionally, the PMTA review time is long, and the reasons for rejection are often procedural (such as document format errors), preventing tobacco products that meet substantive conditions from obtaining marketing permits, which greatly hinders the sale of tobacco products.
3. Transparency of Warning Letter Enforcement Procedures
Some public welfare organizations and parents believe that most small operators only care about profits and may only take down products from their websites while still selling illegal products; the parent group complains about the lack of transparency in the FDA's enforcement of warning letters, specifically regarding whether there is a response to warning letters, suspecting that the FDA is merely enforcing on paper, thus demanding the FDA to enforce strictly and regulate rigorously.
3. Comments from Zheng Mingwei's Lawyer Team
Regarding the FDA's meeting, Zheng Mingwei's team, standing on the position of Chinese e-cigarette companies, has submitted the first comment to the FDA in the Federal Register regarding the relief of warning letters and import alerts. Key points of the comments include:
1. The guidance on how to remove items from the import alert list is unclear;
2. The wording "may be" in the import alert does not clarify whether the product truly lacks marketing permission. Using such formatted language without specific evidence is detrimental to importers' understanding of how to respond to import alerts; companies do not know whether the products are theirs, thus the FDA should increase a relief procedure allowing companies to inquire about the information of the accused products to confirm whether they are their products;
3. The Tobacco Products Center has special internal procedures for enforcing warning letters regarding tobacco products. There are no detailed guidelines informing what evidence and documents should be provided in response to warning letters, and the FDA's enforcement procedures should be transparent and public;
4. Other opinions beneficial to the relief of Chinese e-cigarette companies.
4. Conclusion
The U.S. is the world's largest e-cigarette market, and Chinese e-cigarette companies, as potential main forces for future exports, should understand and comply with U.S. e-cigarette laws and regulations. Therefore, we recommend that all Chinese e-cigarette companies actively participate in such meetings to represent Chinese companies. Comments on this FDA meeting can be submitted to the Federal Register by August 29, 2023, U.S. time. For any opinions that need to be submitted through us, please feel free to contact us; we are duty-bound.
Authors of this article:
Zheng Mingwei, Master of International Law from Shenzhen University, Director of the Comprehensive Business Committee and Equity Partner at Zhongyin (Shenzhen) Law Firm.
Lin Hongping, Master of Civil and Commercial Law from Huazhong University of Science and Technology, Master of Advanced Accounting from The Chinese University of Hong Kong;
Jiang Yubin, Master of Law from the University of Birmingham, UK;
Yang Aoyu, Bachelor of Law from the University of Hong Kong, Doctor of Law from the University of California, Irvine, passed the California Bar Exam.



