China’s first batch of replacement-license e-vaporizer manufacturers emerge, August applications are
License renewal has become a hot topic among many e-cigarette manufacturers in China recently. Especially in Shenzhen and Dongguan, which have a large number of licensed manufacturers and brands.
According to some industry insiders, the license renewal process for the "e-cigarette production license" that expired in June has basically been completed since the renewal began, and the paper licenses will be delivered successively. It was also mentioned that the renewal for July is still being processed in the system. According to regulations, if the license expiring at the end of July is not renewed today, it will be the last day to apply. The renewal for August is also about to begin!
It is reported that to promote enterprise management, relevant departments organized discussions with related companies today (July 14), setting up sub-venues in Shenzhen and Dongguan to lay the groundwork for the August renewal work, showing the government's concern for the vaping industry.

(A meeting was also held in Dongguan)

(August renewal is also coming)
In fact, by the end of June, some companies had officially received new licenses.
Most of the first batch of licensed companies in June last year were e-liquid companies, with about 30 in total, so the first batch of renewals is also primarily successful for e-liquid companies. For example, Huajia Biotechnology has already obtained its license.

(Huajia Biotechnology has obtained its license)
One observed e-liquid company has a license issued on June 30, 2023. Its scope of permission includes both domestic sales and exports.
It is worth mentioning that if there are no other additional changes, the renewal process generally takes about half a month.
Some new highlights have emerged in this renewal process.
1
Has the validity period been extended?
For example, one e-liquid company successfully renewed its license, and the validity period on the license is two years, from July 2023 to the end of June 2025. This is a noticeable extension compared to the one-year validity period of most licenses issued in the first batch last year.

(Validity period has been extended to two years)
However, according to the regulations from June this year, the validity period may be dynamically adjusted based on order conditions, which could be a changing factor. It was specifically mentioned that "the validity period of the license may be dynamically adjusted based on operational conditions."
2
Production scale remains limited.
A license generally includes the company name, address, legal representative, scope of permission, main investors, product brands, validity period, issuing authority, and issuing date, as well as "production scale."
Among them, "production scale" specifically mentions, "not exceeding the nationally approved data," meaning there are limits on production requirements.
3
Brand companies have also obtained licenses.
According to industry insiders, in addition to e-liquid companies, some brand-holding companies have also obtained licenses. This insider stated, "They just went to pick up the paper licenses."

(Some brands have also obtained licenses)
Moreover, these brand-holding companies primarily focus on exports. It is worth noting that last year, it was relatively rare for e-cigarette brand-holding companies to focus solely on exports, but this year, it seems that some brand-holding companies have shifted their business focus entirely to exports, including pod exports, device exports, and a combination of both.
4
Some may be eliminated.
According to one insider, "There may be new qualified production companies added," indicating a potential entry and exit, which may eliminate some companies.
However, the new license application window has not yet opened, and it may take time until the renewal process is completed and statistics are available.
Looking back at the data from last year, by June 30, 2022, a total of 35 e-cigarette-related companies had applied for the issuance of tobacco monopoly production licenses.
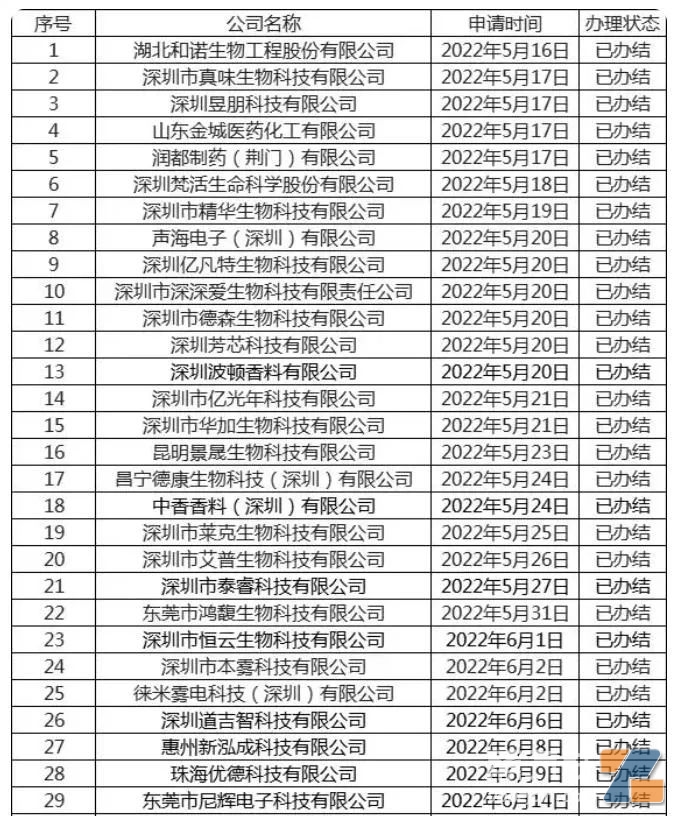
(List of companies that obtained licenses before the end of June last year)
The earliest licensed companies included Hubei Huo Nuo Biotechnology, Zhenwei Biotechnology, Shandong Jincheng Pharmaceutical, Shenzhen Jinghua Biotechnology, Desen Biotechnology, Hongfu Biotechnology, Huajia Biotechnology, Dekang Biotechnology, and other e-liquid companies.
The first batch of companies received their licenses between May 16 and 20.
After June 1, brands such as Benwei Technology and Laimi Vape also obtained licenses. By the end of July, a total of only about 130 companies had obtained licenses, with only over 90 being production companies. In August, companies like Yuan Gelin, Youweier Technology, Klaipeng, and Yueke also obtained licenses. From the list in July, some established leading manufacturers began to obtain licenses, such as Zhuoli Neng, Haoyuan Electronics, Xingke Electronics, Jier Technology, Aivi Pusi, Niwo Network, and Meizhong Lian.
In summary, the renewal process has begun, but this round only allows for "license continuation, modification, and reapplication" for renewal, meaning the new application window is not yet open.
From the requirements, the target is specifically "production companies that were licensed last year." It is particularly mentioned that for renewal with supplementary filling items, resubmission must be done cautiously.
Here are some key points to note:
For example, applications must be submitted 10 days before expiration; applications must be processed through online channels; changes in licenses must be verified on-site, but for license continuations, on-site verification is generally not required; for increasing new production capacity, basic construction or technical transformation approval procedures must be followed and approved; changes in address due to objective reasons must follow the change renewal process, while non-objective changes must follow the reapplication process, and both changes and reapplications require on-site verification and approval; changes in company investors must be evaluated for any illegal activities, and foreign investment regulations are still not allowed.
In addition, transitioning from export to domestic sales will complicate the process, as transitioning from OEM (export) to adding applications for OEM (domestic sales) will require reapplication for licenses and must meet national standards and provide genuine domestic e-cigarette product OEM orders or contracts. Transitioning from OEM (export) to becoming an e-cigarette production company will require providing genuine and legal e-cigarette product registered trademark documents and following the reapplication process.
It is expected that in the upcoming August, hundreds of companies will be busy with renewal matters, continuously contributing to the Chinese vaping industry's overseas expansion. Wishing all manufacturers a timely renewal.



