Australia Tightens Regulation: Banned Ingredients Found in RELX, IGET and Others, Compliance Becomes
Australiae-cigarette marketChanges are taking place.
As early as 2021, Australia has alreadye-cigarettesIt is regulated as a prescription drug, but it was discovered after sorting out the key time points of e-cigarette regulation in Australia. It was not until more than half a year that Australia really tightened supervision:
December 17, 2020: The "Poison Standards" were revised and approved; nicotine was included in the scope of prescription drugs;
May 13, 2021:"Nicotinee-cigarette productsStandard (TGO 110) was adopted;
October 1, 2021: The amendment takes effect; a prescription is required to purchase e-cigarettes, and e-cigarettes are only sold in pharmacies;
October 1, 2021: The Nicotine Electronic Cigarette Product Standard (TGO 110) comes into effect;
May 2022: Mark Butler becomes the new Australia Minister of Health and Aging;
Summer 2022: Mark Butler promotes TGA to conduct public consultation on e-cigarettes;
December 2022: The government proposes to strengthen supervision and enforcement of all e-cigarettes;
May 2, 2023: National Tobacco Strategy 2023-2030;
May 2, 2023:"New Deal in May"-announced the launch of strong supervision and law enforcement on e-cigarettes;
June 13, 2023: First penalty after the "May New Deal";
More than a year after the launch of the "prescription drug model", the Australia e-cigarette market is still in a state of barbaric growth. Until many key events that occurred this year,e-cigarette enterprisesWe are beginning to realize that compliance entry into the Australia market is imminent.
First, the TGA has begun frequent enforcement; for example, on June 13 this year, the TGA announced that it had issued 38 infringement notices to four Sydney companies, with a total amount of AU$588,840, involving 22 batches of imported goods, totaling 379,600 e-cigarettes. The handling was carried out with great speed-after being intercepted by border guards, it was directly seized and destroyed in accordance with TGA recommendations.
The reasons for the penalty officially announced by the TGA: The above-mentioned e-cigarettes were neither registered with the prescription drug ARTG, nor were they found to contain prohibited ingredients listed in TGO 110 in TGA laboratory tests-penalties for double violations have hardly occurred before.
Second,RELXOther brands have been found to contain prohibited ingredients. Test results released by the TGA laboratory showed thatYue Ke relx, a number of products such as IGET have been tested to contain prohibited ingredients. Judging from its Expiry date, these products that were detected for prohibited ingredients obviously entered the Australia market after the release of the Australia e-cigarette product standard:
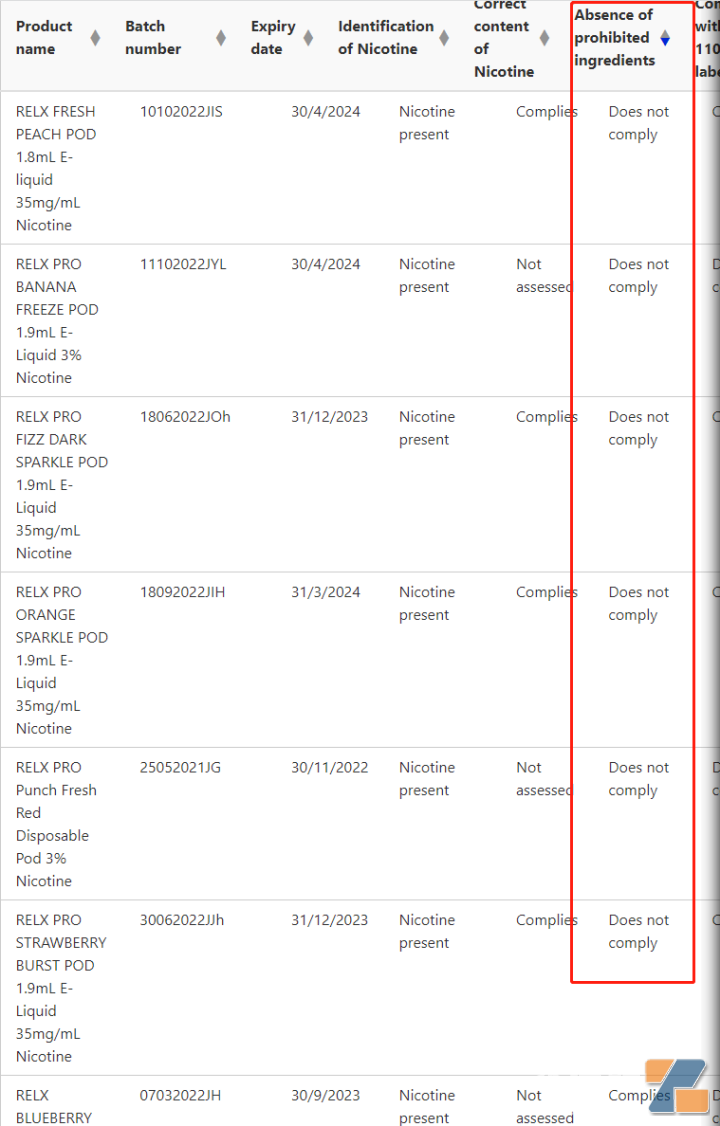
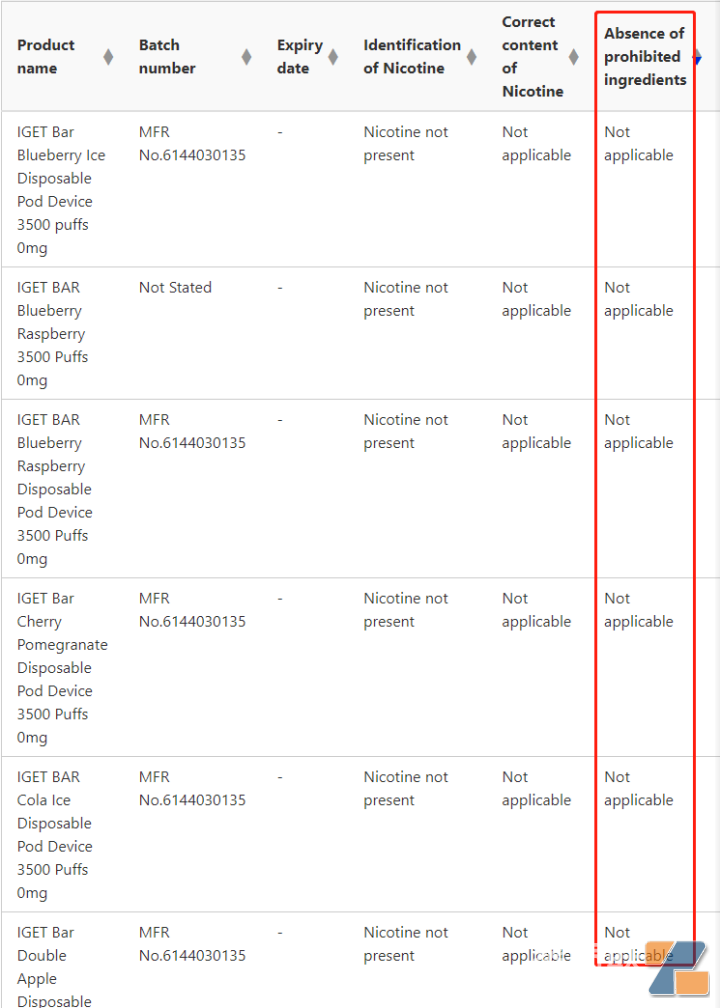
According to Gewu Consumer, no e-cigarette in Australia has registered ARTG prescription drugs. This means that if the latest penalty is imposed on June 13, the aboveyueshiBranded products such as RELX are within the scope of TGA law enforcement.
From this point of view, the compliance of China e-cigarette companies in the Australia market is obviously imminent. So the question arises. What is Australia's regulatory approach to e-cigarettes? And why has there been sudden tightening recently?
Current product standards: crude standards and unobtainable prescription drug registrations
The current Australia e-cigarette product standard "Nicotine E-Cigarette Product Standard (TGO110)" was adopted as early as 2021 and is the "legacy" of the previous Australia Minister of Health and Aging. Since the supervision of e-cigarettes as prescription drugs has been clarified at the end of 2020, Australia's e-cigarette product standards have a strong "drug style", mainly reflected in:

1,smoke oilComposition;
The active ingredient is limited to nicotine only.nicotine saltThe nicotine concentration of free radical tobacco oil should not exceed 100mg/ml. The content or concentration of nicotine must be no less than 90.0% of the stated content and no more than 110.0% of the stated content (false standards are prohibited).
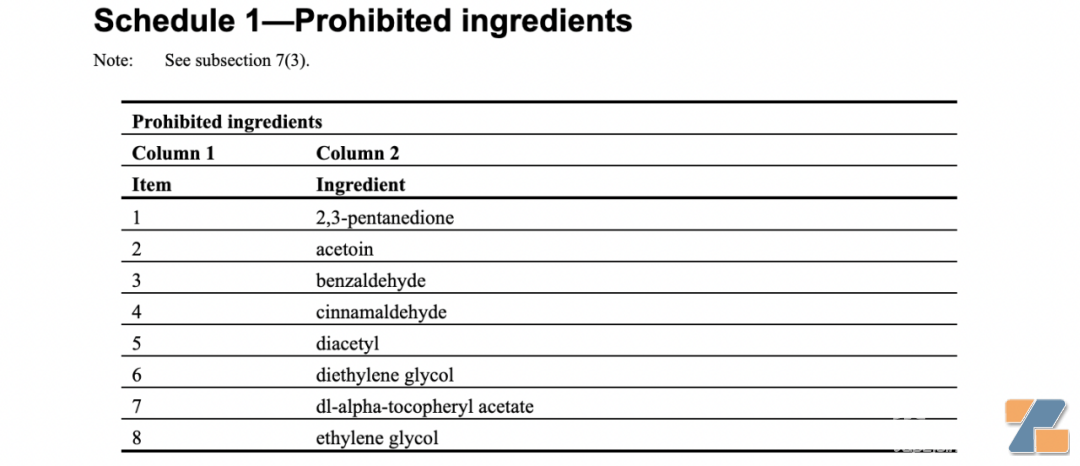
Must not contain the following 8 prohibited ingredients: 2,3-pentanedione, acetoin, benzaldehyde, cinnamaldehyde, diacetyl, diethylene glycol, dl-α-tocopherol acetate, ethylene glycol: #p#Page Title #e#
2. Product label
Must contain content:
Names of all active ingredients (nicotine), other ingredients;
Names of all flavor ingredients;
Nicotine concentration expressed in mg/ml;
Warnings including keeping out of contact with children, avoiding contact with eyes, and avoiding contact with skin.& nbsp;
Product labeling requirements:
Product labeling containing the above information must be provided
Need to be provided in printed, posted or accompanying form on the container or main packaging;
Label information must be written in English, readable, clear and non-ambiguous, and durable.
Under Australia law, prescription drugs must be registered with the Australia Therapeutic Goods Register (ARTG) before they can be legally imported into Australia or supplied in Australia. The conditions are as follows:
Data demonstrating product quality, safety and effectiveness for intended use
Fill out the form;
Payment of fees;
So, which e-cigarettes have completed ARTG registration? Unfortunately, there are none. The logic underlying TGA's supervision of e-cigarettes is the same as that of drugs, which requires a large amount of test data. Currently, all e-cigarettes in Australia are in an "unapproved" state.
Neglected regulation: runaway sales and rampant illegal products
What should Australia's compliant e-cigarette purchase method be? First, after obtaining a doctor's prescription, you can buy it at a pharmacy with the prescription; second, after obtaining a doctor's prescription, you can order online from an overseas retailer. It is recommended that the package sent by the latter also need to come with a prescription to be inspected-the core is, doctor's prescription.
The official explanation is as follows:
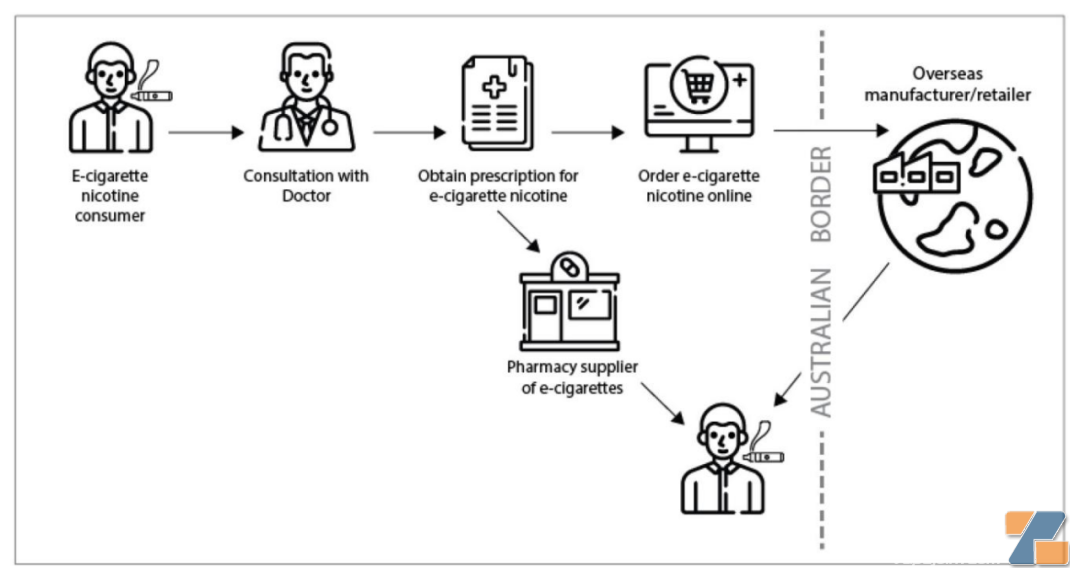
In fact, what about the e-cigarette market in Australia? At the Global Nicotine Forum 2023 held in June this year, Caroline Beaumont, a general practitioner of Australia and founder of MedicalNicotine, shared the chaos in the e-cigarette market in Australia.
She made it clear that the "prescription drug supervision model" that has been launched since 2021 has not actually cracked down on the spread of illegal e-cigarettes. She estimated that illegal e-cigarettes consumption may account for 80% of total e-cigarette consumption, and it is almost entirely consumed by young people. Consumption.
Relevant people concerned about the Australia e-cigarette market also told Gewu Consumer that Australia e-commerce companies that do not have the qualifications to sell e-cigarettes are still openly selling e-cigarettes and have not prompted any prescriptions.
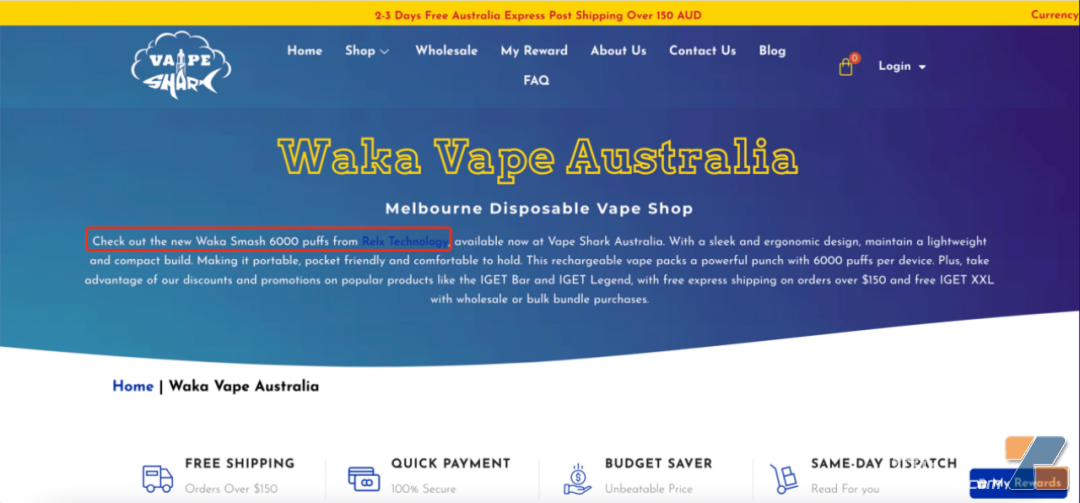
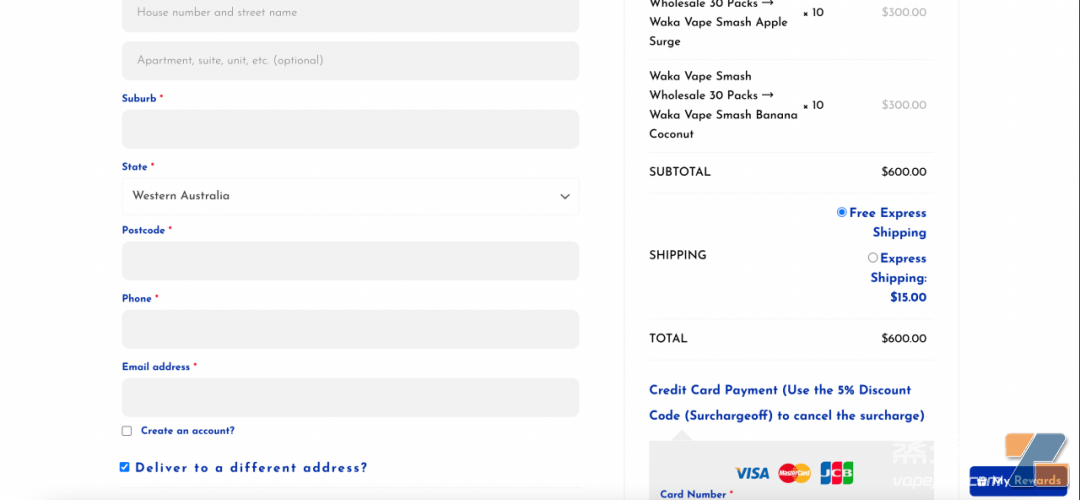
For example, an Australia e-cigarette e-commerce company is still publicly selling Waka brand disposable e-cigarettes, and said that this e-cigarette comes from Relx Technology. Entering the purchase page also lists the current remaining inventory in detail. The entire purchase process is very simple and there are no hints about prescriptions:
Why supervision is suddenly tightened: The new minister's "three fires"
In Australia, e-cigarettes are regulated by the Therapeutic Goods Administration (TGA), which is part of the Australia Department of Health and Aging (DHAC) and is the supervisory authority for therapeutic commodities in Australia. Why have you suddenly clenched your fists in recent months? The reasons behind this involve many complex factors and we will not dwell on them too much. We have summarized two core points that are convenient for everyone to understand.
First, the rapidly growing underage use rate does not match the image of tobacco control;
Mark Butler, Minister of Health and Aging of Australia, once disclosed the following set of data: one in six teenagers aged 14-17 have smoked e-cigarettes, and one in four among those aged 18-24 have smoked e-cigarettes; and when asked more than a thousand teenagers aged 15-17 "where to buy e-cigarettes," four in five said they found it easy or relatively easy to buy e-cigarettes in retail stores.
The current smoking rate in Australia is only 10%, which is driven by measures such as Australia's undecorated tobacco packaging, which were the report card handed over by Mark Butler's Labor Party during its previous administration.
Second, the "three fires" of the new generation of regulatory authorities vow to rebuild their glorious achievements;
After the Labor Party won the 2022 Australia federal election, Mark Butler became Minister of Health and Aging. What he faced after taking over the position was the mess left by his predecessor: a former minister tried to tighten border controls, allowing only individuals to import non-nicotine e-cigarettes. But e-cigarettes have since gotten out of control, and a large number of "false" e-cigarettes containing nicotine have entered the market.# p#pagination title #e#
It was very easy for e-cigarettes to enter Australia before. On the one hand, they could take advantage of the loophole of Australia regardless of zero nicotine and directly falsely label nicotine; on the other hand, e-cigarettes are small in size and can also muddle through false declarations.
Mark Butler will consult with health organizations and communities in the summer of 2022 to plan the direction of e-cigarette regulation in Australia. One of the driving forces behind this is the hope of recreating the previous record of tobacco control during the Labor Party's administration.
Regulatory changes: New Deal and Lightning Action in May plunged the Australia market into unknown
Faced with an out-of-control market, on May 2 this year, Mark Butler officially announced the "May New Deal"-Australia launched strong supervision and enforcement of e-cigarettes, covering 2023-24, with a total budget reaching A $737 million, measures include:
Stop importing non-prescription e-cigarettes;
Improve minimum quality standards for e-cigarettes, including restricting taste, color and other ingredients;
E-cigarettes need to be packaged similar to drugs;
Reduce the permitted nicotine concentration and volume;
Ban all disposable e-cigarettes.
Although the laws corresponding to the above measures are still being formulated, we can still interpret a new direction from them-in order to reproduce the Labor Party's previous glorious record of tobacco control, Mark Butler is breaking and reshaping previous Australia regulatory measures, which is reflected in:
Completely kill it once;
Strictly crack down on e-cigarettes that have not registered ARTG;
TGO110 may be obsolete and new product standards will be developed;
E-cigarettes are moving closer to drugs in Australia;
Australia states responded quickly and positively. For example, on July 11, South Australia launched an eight-week enforcement blitz against the illegal sale of nicotine e-cigarettes. At present, e-cigarette regulation in Australia has actually entered a "transition period"-the general direction has been set, but detailed regulations are still being formulated. However, states have begun to strengthen supervision of e-cigarettes according to their own understanding.
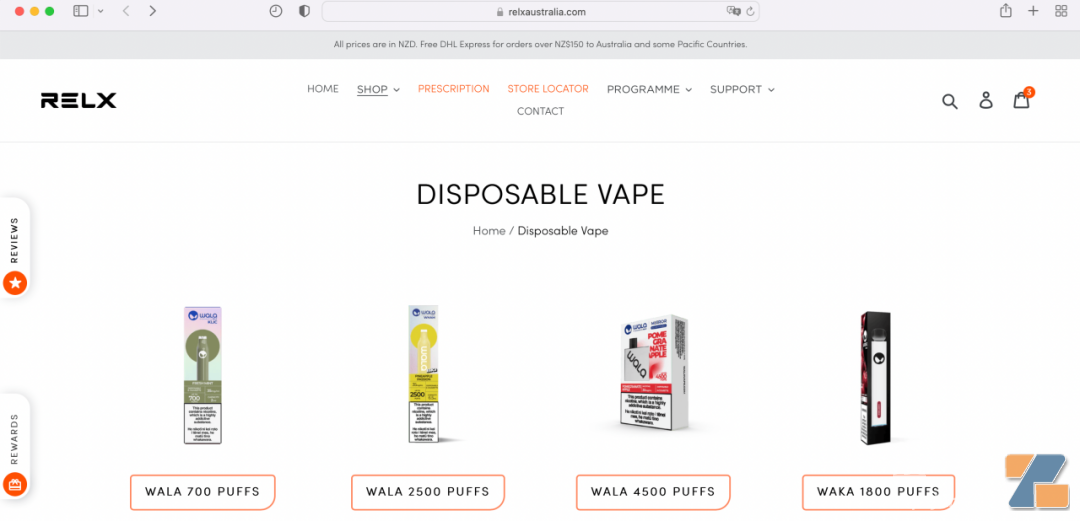
However, according to Gewu observation, although Australia's attitude towards cracking down is quite clear, there are still brands trying to continue selling every minute of the current window. For example, Yuke's relxaustralia.com still sells waka Pacific brand WALA and mentions that "all prices are in New Zealand dollars. Australia and some Pacific countries can enjoy free DHL express services for orders exceeding New Zealand dollars."-- Although it is impossible to determine whether this is the behavior of a local dealer, it is obviously inconsistent with the new Australia regulations.
Reshuffle is imminent: Big Tobacco's profits are exchanged for the market, and the channel's voice is further enhanced
Interestingly, Australia's tobacco industry, which had tried hard to draw a clear line, staged a wave of "home theft." On July 6, the Guardian reported that Philip Morris International reached an agreement with Australia pharmacies to supply e-cigarettes at a price below cost.
Reports say that as long as pharmacists sign a supply agreement directly with Philip Morris International, they can obtain supplies at a profit margin of 80%. But pharmacies are not allowed to sell VEEVs for more than $14.90the cartridge, e-cigarette equipment may not be sold for more than $19.90.
The suggested retail price of similar cigarette bomb products is generally US$24.99-in other words, PMI is trying to grab the market through high gross profit and low selling prices, and many pharmacies have already signed agreements.
PMI's move is more like a rainy day and does not necessarily immediately overturn the pattern of the Australia e-cigarette market. But on the other hand, as Australia is already formulating new product standards and regulatory measures, the voice of pharmacies as the only channel is expected to be further enhanced. How to do business with pharmacies will become a new question in the next stage of the Australia market.



