Racing Toward PMTA: The Uncertain Fate of Chinese Manufacturers in the US
 In the first month of 2024, the FDA has successively conducted SMOK,suorin、BLU PLUS、Bidi sticke-cigarette brandsMarketing Denial Orders (MDOs) have been issued on some products of the company, which means that these companies are not allowed to market or distribute these products in the United States or risk FDA enforcement action. The last time the FDA issued an MDO was in October 2023. Two months later, it issued MDOs intensively. What did the FDA's move mean before the largest TPE exhibition in the United States? This will be the most important thing for the United Statese-cigarettesIs the beginning of a new round of supervision?
In the first month of 2024, the FDA has successively conducted SMOK,suorin、BLU PLUS、Bidi sticke-cigarette brandsMarketing Denial Orders (MDOs) have been issued on some products of the company, which means that these companies are not allowed to market or distribute these products in the United States or risk FDA enforcement action. The last time the FDA issued an MDO was in October 2023. Two months later, it issued MDOs intensively. What did the FDA's move mean before the largest TPE exhibition in the United States? This will be the most important thing for the United Statese-cigarettesIs the beginning of a new round of supervision?
[Two Supremes Original] The U.S. Food and Drug Administration (FDA)'s supervision of atomized e-cigarettes seems to be tightening again in 2024, but some China manufacturers are still waiting for their own "ticket."
In the first month of 2024, the FDA issued marketing refusal orders (MDOs) on some products of the SMOK, Suorin, BLU PLUS, and Bidi stick e-cigarette brands, which means that these companies are not allowed to market or distribute these products in the United States, otherwise they will face the risk of FDA enforcement action. The last time the FDA issued an MDO was in October 2023. Two months later, it issued MDOs intensively. What did the FDA's move mean before the largest TPE exhibition in the United States? Will this be the beginning of a new round of regulation of e-cigarettes in the United States?
 Four MDOs issued by the FDA in 2024| Source: FDA
Four MDOs issued by the FDA in 2024| Source: FDA
Four brands, belonging to three categories
The two leaders conducted an inventory of the MDO products issued by the FDA since January and found that the products of four brands involve three categories: open e-cigarettes, replaceable e-cigarettes, and disposable e-cigarettes. Among them, there are both China companies and British companies. The FDA's actions seem to be deliberately "naming" all categories of atomized e-cigarettes at the beginning of 2024. Moreover, these products are well-known in their respective types and are not small brands or manufacturers that no one knows.
January 16, 22 models of SMOKe-cigarette productsThe FDA issued a Marketing Rejection Order (MDO), and the products rejected included smoking equipment, cigarette warehouses,atomizerandthe cartridge。After two top statistics, it was found that the 22 products that SMOK "rejected" this time belonged to six series, all of which were open e-cigarette products. The following is a specific classification list:
SMOK OSUB ONE Series:
- SMOK OSUB ONE Device
- SMOK OSUB ONE RPM Cartridge
- SMOK OSUB ONE RPM Cartridge 3 Pack
SMOK RPM Series:
- SMOK RPM DC 0.8 Ω MTL ATOMIZER
- SMOK RPM DC 0.8 Ω MTL ATOMizer 5 Pack
- SMOK RPM 40 Device
- SMOK RPM Empty Standard Cartridge
- SMOK RPM Empty Nord Cartridge
- SMOK RPM Mesh 0.4 Ω Atomizer
- SMOK PRM 2 Mesh 0.16 Ω Atomizer
- SMOK RPM Mesh 0.4 Ω Atomizer
SMOK Nfix series:
- SMOK Nfix Device
- Nfix DC 0.8 Ω MTL Pod
SMOK POZZ Series:
- SMOK POZZ Device
- SMOK POZZ DC 0.8 Ω Pod
SMOK Nord series:
- SMOK Nord DC 0.8 Ω MTL Atomizer
- SMOK Nord 2 Device
- SMOK Nord 2 RPM Cartridge
- SMOK Nord 2 Nord Cartridge#p#pagination title #e#
SMOK SCAR-P3 series:
- SMOK SCAR-P3 Device
- SMOK SCAR-P3 Empty RPM 2 Cartridge
- SMOK SCAR-P3 Empty RPM Cartridge
 SMOK OSUB ONE set| Source: SMOK
SMOK OSUB ONE set| Source: SMOK
Only three days later, on January 19, the FDA sent another report to ShenzhenNiwo Internet TechnologyShenzhen YouMe InforMATion Technology Co. Ltd.) two Suorin e-cigarette products and Imperial Tobacco subsidiary FoSeveral e-cigarette products from ntem US, LLC's blu PLUS+ have issued refusal to sell orders (MDO).
Among them, Suorin e-cigarette products are Suorin Air and Suorin Air empty refillable cartridge, which are smoking sets and empty cigarette cartridges respectively. From the product introduction, Suorin Air is a product that can be filled with oil; and blu PLUS+ is a "rechargeable disposable e-cigarette."
 Suorin Air| Source: Suorin
Suorin Air| Source: Suorin  blu PLUS+? Carolina Bold 2.0% |Source: blu
blu PLUS+? Carolina Bold 2.0% |Source: blu
However, the FDA's action did not end. Three days later, on January 22, the FDA issued an MDO to Bidi Stick Classic e-cigarettes from Bidi Vapor LLC. According to the official website, Bidi Stick Classic is a disposable product.
 Bidi Stick Classic| Source: Bidi Stick
Bidi Stick Classic| Source: Bidi Stick
Companies that proactively seek compliance
FDA onnew tobaccoThe slow review of the company has always been criticized. As of March 2023, there are only 23 e-cigarette products that have passed PMTA. There are still a large number of e-cigarette products that have not yet received preliminary results from the FDA.
The FDA has designed a set of complex but targeted reviews for PMTA. All products submitted to companies must meet these reviews. Two of the procedures are described on the FDA's official website. The following are the streamlined steps:
Pre-submission meeting: A voluntary formal meeting between the applicant and the FDA to discuss PMTA submissions in the Tobacco Products Program;
Acceptance review: Administrative review to ensure that the product is placed under the jurisdiction of the Tobacco Products Center and confirms that the legal and regulatory requirements of the application are met;
Submission for review: The threshold for determining whether the application contains sufficient information to allow substantive review;
Substantive review: FDA's assessment of scientific information and data in the application, certification of output deficiencies: Additional information is needed to make a marketing authorization decision. Applicants have 180 days to respond to such letters;
Review results: Marketing order issued or marketing order rejected.
Postmarket reporting: Applicants are required to establish and maintain FDA records necessary to make reports to determine or facilitate a determination of whether there is reason to withdraw or temporarily suspend market-approved orders.
.png) review link| Source: FDA
review link| Source: FDA
For example, the first SMOK to be issued an MDO in 2024, its official website data shows:
In July 2020, it submitted a Pre-Market Tobacco Product Application (PMTA) to the FDA for its Electronic Nicotine Delivery System product
In August 2020, SMOK received a letter of acceptance for premarket application for tobacco products from the US Food and Drug Administration;
- #p#Pagination Title #e#
In September 2020, SMOK passed the second round of pre-market applications for tobacco products and entered the final stage.
 SMOK's statement submitted to PMTA| Source: Two supremacy
SMOK's statement submitted to PMTA| Source: Two supremacy
At the time of submission of the PMTA, SMOK's market share in the U.S. still remained at a high level. According to data from the 2021 National Youth Tobacco Survey released by the FDA and the Centers for Disease Control and Prevention (CDC), 9.6% of teenagers said they use S.MOK Products, andJUULRanked next, with a proportion of 5.7%.
 National Youth Tobacco Survey, United States, 2021| Source: CDC
National Youth Tobacco Survey, United States, 2021| Source: CDC
Three and a half years after submitting the PMTA application, on January 16, SMOK finally received the news that it was "rejected", but Shenzhen, the company owned by the SMOK brand.AvipusTechnology Ltd.(IVPS) has announced that it has filed an appeal to the U.S. Court of Appeals for the Fifth Circuit against the U.S. Food and Drug Administration (FDA)'s marketing denial order for its SMOK-branded open e-cigarette kits and related spare parts.
 SMOK says it will sue FDA| Source: SMOK
SMOK says it will sue FDA| Source: SMOK
According to public information, SMOK is also one of the six China companies that submitted PMTA before September 9, 2020e-cigarette enterprisesOne thing worth noting is that Suorin, a subsidiary of your and my group, is also among them and was also issued an MDO this month.
In the same batch are Simmore's own tobacco brand VAPORESSO, e-cigarette supply chain Heyuan Group, Geer's tobacco brand VOOPOO, and Boulder.
Suorin, who was also issued an MDO by the FDA, announced the PMTA application process in the IECIE 2020 issue on August 20, 2020. Suorin said that the application materials submitted before May of that year were "confident in the content and quality." Suorin said the PMTA application has been submitted including the results and summaries of HPHC testing, extractables and extractables testing, stability studies, toxicological analyses, clinical trials and perception studies.
 Suorin's explanation at IECIE 2020| Source: IECIE 2020
Suorin's explanation at IECIE 2020| Source: IECIE 2020
In addition to the two companies that were explicitly rejected, there are four e-cigarette manufacturers whose PMTA is pending, waiting for the FDA's response, which is the release stage of the review results. After reviewing relevant information, the two supremacy sorted out their application status before January 25, 2024 as follows.
JierIts open e-cigarette brandVOOPOOThe official website shows:
The PMTA was officially submitted on August 27, 2020, U.S. time and passed the first round of review;
Passed the first round of review on September 5, 2020;
The second round of review was passed on September 30, 2020.
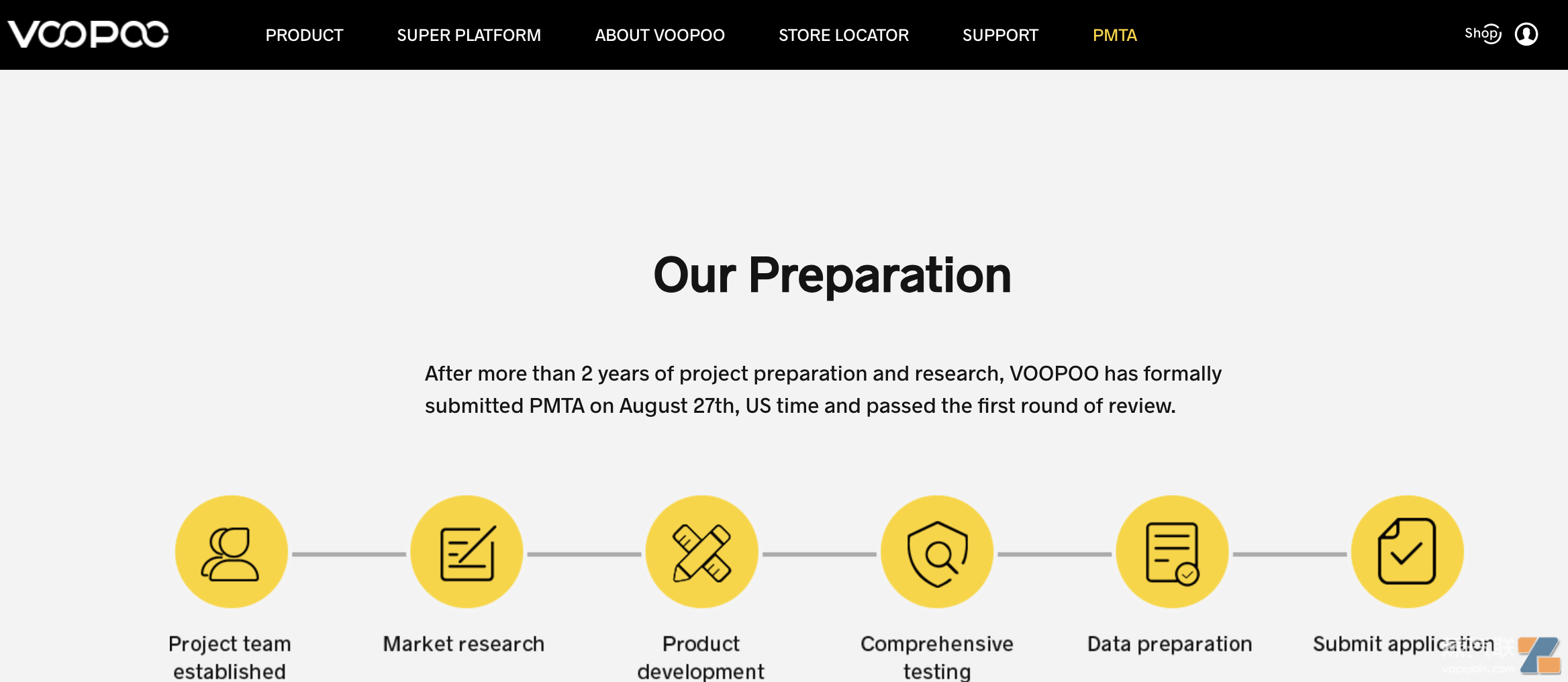 VOOPOO official website introduction to PMTA progress| Source: VOOPOO
VOOPOO official website introduction to PMTA progress| Source: VOOPOO
According to VOOPOO, it "spent 100 million yuan in the entire project process and took 20 months from preparation to application to complete PMTA testing requirements such as product composition analysis, manufacturing research, and non-clinical and clinical human studies."
Currently, the application for VOOPOO is still under review by the FDA.
BoulderIs the only one submitted simultaneouslysmoke oilWith the China manufacturer of equipment, it submitted PMTA for six types of tobacco fluids and three types of equipment to the FDA on September 8, 2020. It is reported that this is the first China company to submit PMTA of e-cigarette fluid in the United Statese-cigarette companies。It has passed the second round of review and entered the stage of substantive scientific review. A manuscript published by Boulder on January 21, 2022 mentioned,"#p#pagination title #e#bodeIt is the only Chinese-funded company that has entered the substantive scientific review stage of PMTA."
Judging from the report of Vaporvoice on October 3, 2020, Boulder will only include four devices in the initial application, two devices: Boulder Rock and Aspen Slim, and two flavors of e-cigarette fluids: American Blend and Menthol.
 Boulder's introduction to the progress of PMTA| Source: Boulder
Boulder's introduction to the progress of PMTA| Source: Boulder
Similar to VOOPOO, Boulder's application has not yet received a final response from the FDA.
As an e-cigarette supply chain company in China, Simmore International andfirst UnionPMTA applications are more relevant to cooperative enterprises.
According to the regulations of the U.S. FDA, customers and foundries need to jointly submit PMTA certification to the FDA. For example, when a foundry company cooperates with customers to apply for PMTA, it needs to provide samples and documents, and the factory also needs to pass inspection. If a customer needs to change the OEM, his product needs to be re-applied through PMTA. For the above reasons, once a product is certified, the foundry will not be easily changed.
One of the domestic supply chain giantsHeyuanThe group announced that it received the FDA acceptance letter on August 25, 2020. the notice will firstunionThe submitted materials were moved to the substantive review stage of the PMTA process; on September 8, 2020, Heyuan Group completed the second round of review of the PMTA application and received a filing letter from the FDA.
 Heyuan Group received PMTA filing letter| Picture source:VAPTIO
Heyuan Group received PMTA filing letter| Picture source:VAPTIO
Heyuan Group once stated that before entering the substantive review stage, after more than a year of hard work, it completed more than 100,000 pages of application materials and evidence to support its claims, as well as multi-dimensional test data and reports to prove that the company's products are beneficial to public health.
According to the two supreme understandings, Heyuan Group is the only two e-cigarette factory in China that has been reviewed on-site by the FDA, and the other is Simore.
As an OEM company, Simmore has received PMTA for many of its OEM products. According to the 2023 mid-year report released by Simmore,"The FDA has issued marketing authorizations for 11 closed-system tobacco-flavored ENDS products produced by the Group for customers”。At the same time, Simmore International also provides its own open e-cigarette brandVAPORESSOApply for PMTA.
On August 20, 2020, VAPORESSO's first round of PMTA application was accepted by the FDA;
On September 1, 2020, the first batch of PMTA products submitted by VAPORESSO have passed the second round of FDA filing;
On March 12, 2021, Vaporesso responded to the FDA defect letter;
On April 8, 2021, Vaporesso completed the FDA remote regulatory assessment.
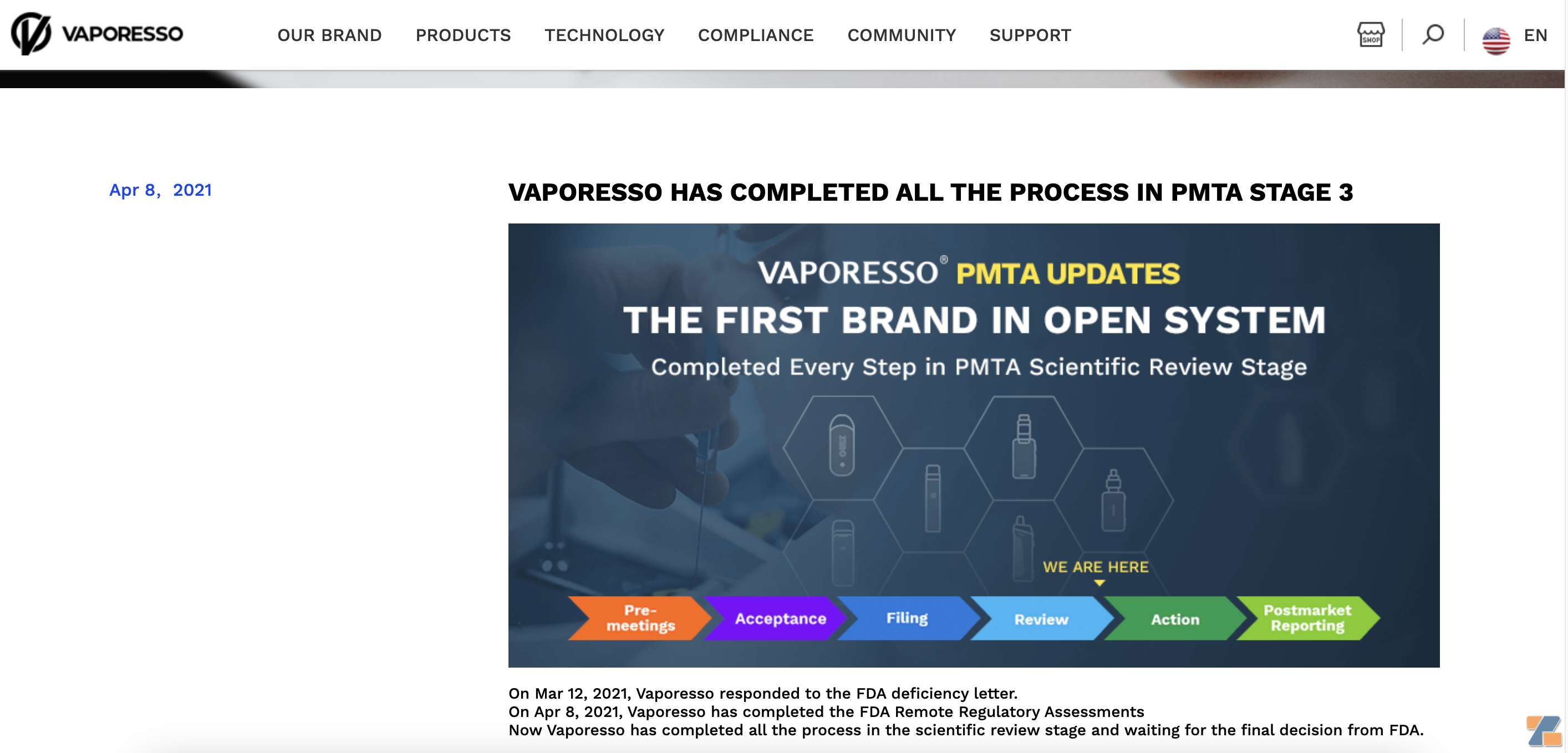 Introduction to Vaporesso PMTA| Source: Vaporesso
Introduction to Vaporesso PMTA| Source: Vaporesso
Vaporesso's official website shows that it has completed all processes during the scientific review stage and is waiting for the FDA's final decision.
Looking back at the sea tide in 2020
The above companies only took stock of six China e-cigarette companies that submitted PMTA before September 9, 2020. Domestic companies may want to go to the United States to obtain PMTA, but in fact it is a change in the domestic and international situation.
On November 1, 2019, the State Tobacco Monopoly Administration and the State Administration for Market Regulation jointly issued the "Notice on Further Protecting Minors from E-Cigarettes"(hereinafter referred to as the "Notice"): Urge e-commerce platforms to close e-cigarette shops in a timely manner and remove e-cigarette products from shelves in a timely manner; urge e-cigarette production and sales companies or individuals to close e-cigarette Internet sales websites or clients in a timely manner; urge e-cigarette production, sales companies or individuals to withdraw e-cigarette advertisements posted through the Internet.# p#pagination title #e#
 The State Tobacco Monopoly Administration and the State Administration for Market Regulation jointly issued the "Notice on Further Protecting Minors from E-Cigarettes"| Picture source:
Tobacco Bureau website
The State Tobacco Monopoly Administration and the State Administration for Market Regulation jointly issued the "Notice on Further Protecting Minors from E-Cigarettes"| Picture source:
Tobacco Bureau website
China's online supervision of e-cigarettes has since been followed by laws, but at this time many companies are already anchored in the sea, looking at the United States on the other side of the ocean.e-cigarette industryJUUL, the most promising rising star, is gradually declining. One month before China issued the "Notice", on October 18, 2019, JUUL announced that it would stop selling flavored e-cigarettes in the United States, retaining only tobacco and mint flavors. A super "unicornThe temporary exit of "left opportunities for other companies in the market.
 JUUL announced that it would stop selling flavored e-cigarettes in the United States| Source: CBS
JUUL announced that it would stop selling flavored e-cigarettes in the United States| Source: CBS
In January of the following year, the FDA promulgated a new policy for e-cigarettes in the United States, banning flavored e-cigarette cartridges and retaining only the two flavors of tobacco and menthol. Being ranked first in the worlde-cigarette marketThe United States marched. As a necessary certificate for legal sales, PMTA seems to have become a ticket before going to sea.
 The FDA promulgated a new policy for e-cigarettes in the United States, banning flavored e-cigarette cartridges| Source: FDA
The FDA promulgated a new policy for e-cigarettes in the United States, banning flavored e-cigarette cartridges| Source: FDA
In 2020, China's e-cigarette companies were still seeking how to obtain PMTA. At that time, the industry expected that "the strict standards of PMTA will also usher in a new round of reshuffle in the overseas e-cigarette market." However, few people have noticed the rise of disposable e-cigarettes around the world. These e-cigarettes, which have extremely low barriers to use, are easy to smoke and can be discarded at any time, have conquered consumers in the United States and around the world in just a few years.
The results of the 2023 U.S. National Youth Tobacco Survey show that the brands most commonly reported among current e-cigarette users are: ELFBAR (56.7%), Esco Bars (21.6%), Vuse (20.7%), JUUL (16.5%) and Mr. Fog(13.6%)。
As mentioned above, the e-cigarette brand JUUL had a market share of 76% at its peak in 2018. The latest Nielsen convenience store data shows that Juul's market share dropped from 24.3% to 24.2%, ranking second in the market, and first place is British American Tobacco's VUSE.
 The e-cigarette brand JUUL had a 76% share at its peak| Source: ncbi
The e-cigarette brand JUUL had a 76% share at its peak| Source: ncbi
But neither VUSE nor JUUL are real "winners". Nielsen's report mainly covers large chains, so Renault Tobacco and Altria estimate that these illegal products account for about half of the U.S. e-cigarette market.
The vast majority of these are disposable products. According to industry data held by analyst firm Circana, more than 11,500 e-cigarette products are currently sold in U.S. stores, a 27% increase from 9,000 in June.
The FDA currently has only issued marketing licenses for 23 e-cigarettes, which means that 99% of the atomized e-cigarettes on the market are illegal products under official laws. The FDA will stop passing new aerosolized e-cigarette products in 2023, and this number will remain at 0 throughout 2023.
On October 12, 2021, Vuse Replacement Cartridge Original 4.8% G2 became the first aerosol e-cigarette ever to be applied through PMTA. On June 10, 2022, NJOY DAILY Rich Tobacco 4.5% passed PMTA, becoming the last aerosol e-cigarette product to obtain an MGO (Marketing License Order) as of January 2024.
 The FDA currently only issued marketing licenses for 23 e-cigarettes| Source: FDA
The FDA currently only issued marketing licenses for 23 e-cigarettes| Source: FDA
However, the former belongs to a brand owned by Renault Tobacco acquired by British American Tobacco, while the latter was announced by Altria to be acquired on March 6, 2023. A total of 6 NJOY products have obtained PMTA approval from the FDA.
China manufacturers have not yet issued an MGO for e-cigarette products that have passed the PMTA review. Ironically, China's e-cigarette products account for the vast majority of the U.S. market, but they are "in name", and the legal status of the companies applying for PMTA is still pending.# p#pagination title #e#



