JUUL Sues NJOY for Patent Infringement, Seeks Ban on NJOY Vape Sales
Last Friday, JUUL Labs filed a lawsuit with the U.S. International Trade Commission (ITC) and a federal district court, accusing NJOY Ace vape of infringing on Juul's patents. The lawsuit involves NJOY and its new parent company, Altria Group.
Juul is requesting the ITC to investigate these patent claims and is asking the commission to block the import and sale of "infringing products" in the U.S.
The exact patent infringement allegations are currently unclear, but a summary of the lawsuit filed in the U.S. District Court for Arizona shows that Juul Labs and VMR Products (a Juul subsidiary) accuse NJOY of infringing on five patents related to the "Juulpod" pods.
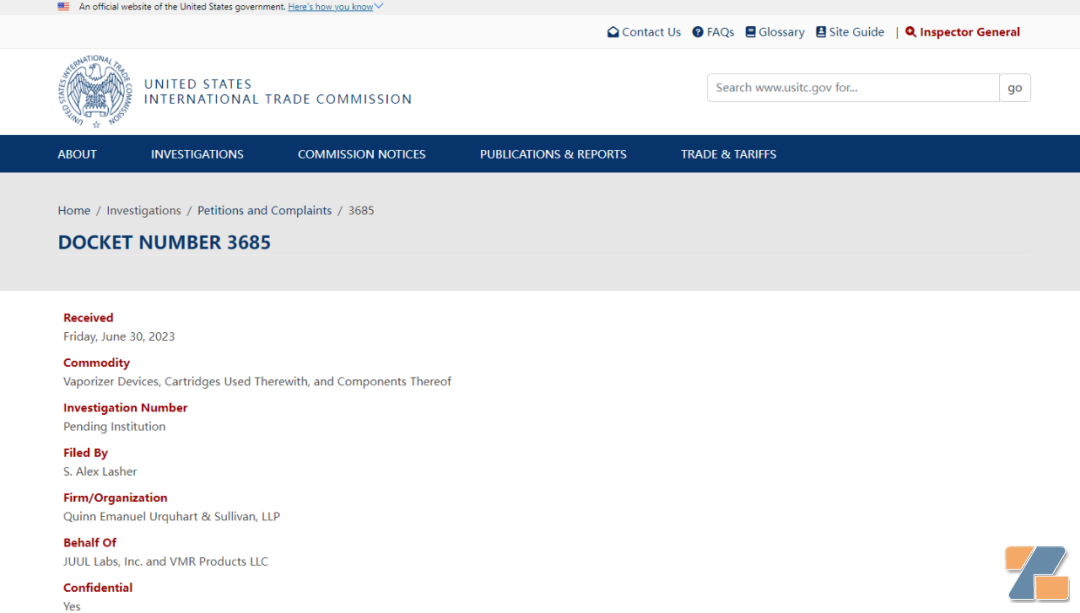
According to the ITC's case files, Juul Labs has requested an investigation into certain atomizer devices, the pods they use, and their components.
It is understood that NJOY Ace is a pod-based vape that belongs to the same category as Juul Labs' Juul device, both using pre-filled pods containing nicotine e-liquid. However, unlike Juul, NJOY Ace and its three tobacco-flavored pods received FDA marketing authorization in April 2022, while Juul's vape has not.
Despite NJOY Ace receiving FDA authorization, its sales have not been very strong, with market share lagging far behind Juul and Vuse Alto. However, the FDA's approval has made NJOY a target for Altria Group, which acquired NJOY for $2.75 billion just three days after selling its stake in Juul Labs.
In addition to NJOY Ace and its three tobacco-flavored pods receiving FDA marketing authorization, the company also obtained marketing authorization for two versions of the disposable vape NJOY Daily, which is the first disposable product to receive FDA marketing authorization.



