For Altria, FDA Approval of IQOS Would Be a Major Opportunity and Turning Point
Philip Morris International submitted its modified risk tobacco product application for the electronic heated tobacco product IQOS to the U.S. Food and Drug Administration on December 5. This aligns with the company's goal set in 2016, and it is expected that the FDA will take at least 60 days to complete its administrative review of the application. Once IQOS progresses, Altria Group will gain exclusive rights to sell these products in the U.S. Altria and Philip Morris have been investing in reduced-risk tobacco products. In 2015, these companies signed a strategic agreement under which PMI would promote Altria's MarkTen e-cigarettes globally, while Altria would distribute PMI's heated tobacco products in the U.S. The companies have also decided to collaborate on regulatory matters concerning these products. In this regard, the two companies are working together to develop MRTP claims for IQOS.
According to Reuters, since the introduction of new laws, Philip Morris is the first company seeking U.S. approval to market tobacco products as less harmful than traditional cigarettes. Once the MRTP claims are approved by the FDA, the company will have a significant market advantage over other reduced-risk tobacco products, including e-cigarettes, which are not allowed to make such claims. The company believes that because its product heats tobacco rather than burning it, it will be safer for consumers. The product has already been available in several test markets. Philip Morris launched the e-cigarette device IQOS in Nagoya, Japan, in November 2014. Since then, the product has been launched in several regions, including Italy and Switzerland. Japan can be considered a key area for the company as it is the only country where the device is fully promoted.
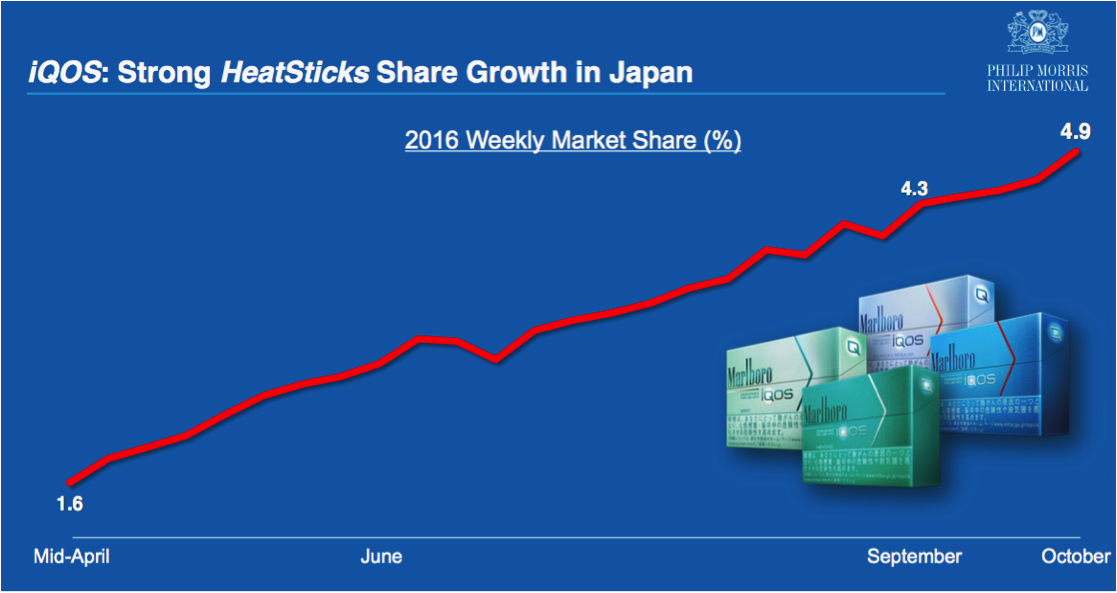
IQOS has seen tremendous growth in Japan
Since its launch, IQOS has witnessed remarkable growth in Japan, with its market share steadily increasing. In 2015, IQOS expanded its distribution in Japan, reaching 60% of the adult smoking population by the beginning of the second quarter. In the third quarter, the market share of HeatSticks grew to 3.5%, an increase of 1.3 percentage points from the second quarter. Additionally, the share reached 4.3% in the last week of September in Tokyo, even peaking at 7.3%, despite limited expansion due to supply constraints. According to the latest data provided by the company, the weekly market share in October increased to 4.9%. Furthermore, the internal product cannibalization rate has decreased from 40% in the early stages to 35% in the second quarter.
Why IQOS will shake the U.S. market
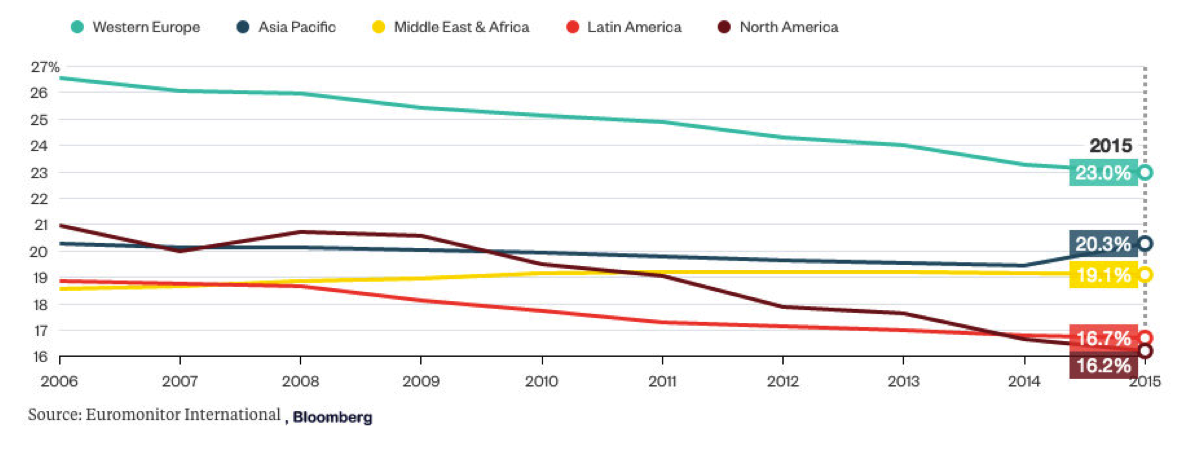
Since the mid-1960s, smoking rates in the U.S. have been declining due to tax increases, bans on tobacco sales, and smoking restrictions in public places, as well as increased consumer awareness. The smoking rate has dropped from 21% in 2005 to below 17% today, and it is expected to decline at a rate of 3% annually before 2040. Additionally, some states in the U.S. are considering raising cigarette taxes in the coming months. This will put increasing pressure on companies like Altria Group to raise the prices of their tobacco products. These initiatives will further impact the number of cigarettes sold by tobacco companies. In the face of these challenges, reduced-risk products in their portfolios will help ensure long-term success.
Currently, available cigarette alternatives are concentrated on liquid-based e-cigarette products that produce vapor and do not contain tobacco. Few of these products can provide the same tobacco experience for tobacco users. In this regard, since IQOS uses tobacco, it may attract these users, and considering that Philip Morris may be the first major company in the industry to obtain FDA approval, this will lead to significant benefits for PMI and Altria.



