E-cigarette report series II | U.S. market: competition continues, consolidation accelerates
report summary
What is the significance of the current review?
The open and dark lines of market competition?Review the sales structure of historical products andFlavor characteristics are the open line that drives every round of market adjustment, and the dark line behind it is the regulatory value orientation centered on protecting minors.。It is mainly reflected in the following: before the official introduction of the taste ban, the PMTA deadline was several times, the regulatory environment was relatively loose, and the flavored and replaceable e-cigarettes represented by JUUL contributed to the main increase in the market. The issue of use by minors aroused social concern, promoted a taste ban, flooded the market with disposable products in gray areas of regulation, and some brands used synthetic nicotine in the intention of further circumventing regulation. Then, the Synthetic Nicotine Act was introduced to fill the loopholes. In the past, the market was a "cat-and-mouse game" of regulation and product strategy.# p#pagination title #e#
tasteThe impact of the ban on markets?From a quantitative perspective, the FDA taste ban came into effect in February 2020, and the shipment level of ammunition replacement products has remained stable since then. There are two reasons behind this: 1) There has been no decline, which is because the FDA has advanced supervision of the head brands of JUUL representatives, fully conveying the "policy trend", and market participants first corrected their business strategies. 2) There has been no increase. The flavor ban gives priority to law enforcement on bomb-changing products, and disposable e-cigarettes have taken advantage of the trend to divert potential customers. It must be acknowledged that seasoning products are an important driver for the industry to attract potential customers and expand its scale.The rise and fall of JUUL also reflects the "blessing and woe interdependence" of flavor characteristics, youth penetration and supervision and law enforcement。In the long run, the taste ban will be of great benefit to society in recognizing the positive value of the industry and stabilizing industry growth expectations.
The direction of the market pattern deduction?Synthetic nicotine has been brought into regulation, flavor differences have narrowed, and marginal changes in share have driven a gradual switch to a comprehensive product power characterized by harm reduction that is jointly determined by brands and manufacturers. With the end of PMTA approval and law enforcement actions becoming the focus of supervision, one-time products have been withdrawn from the market one after another, and consumers benefiting from the bullet-replacement market have returned. At the same time, the FDA has strictly set up reviews to promote secondary clearance of internal supplies. The improvement of penetration driven by harm reduction is a long-term logic. At present, we are more optimistic about the optimization of the competitive landscape in the medium term.#p#Pagination Title #e#
simoreInternational:Binding head brands is expected to fully benefit from industry integration. As the core supplier of VUSE and NJOY, leading brands in the U.S. market, Simmore International continues to deepen R & D and manufacturing and strengthens cooperation and binding with major customers in compliance operations. In the past, revenue growth in the United States was disrupted by illegal operations of one-time products. In the future, it is expected to benefit from the internal integration of one-time product clearance and replacement products, share the market expansion dividends of downstream customers, and the flexibility of manufacturing share is worth looking forward to.
Risk warning:Supervision efforts have not been advanced as expected, menthol products have not been reviewed, etc.。
01#p#Pagination Title #e#
Review:The dynamic game of regulatory environment and business strategy behind the rise and fall of JUUL
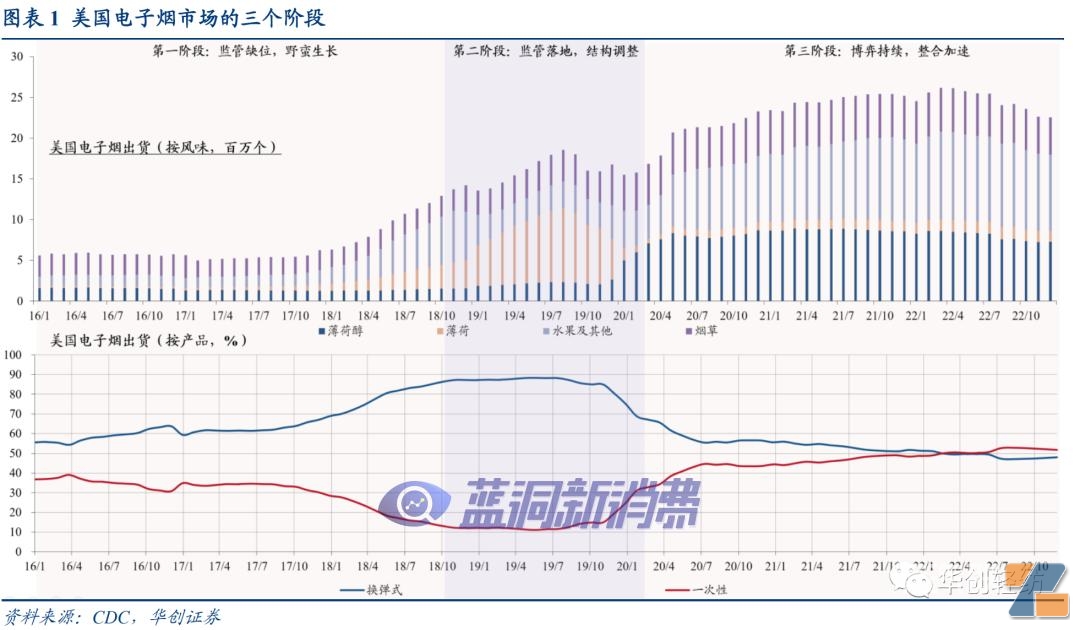
( 1) Overview:Three stages of market interpretation
#p#Pagination Title #e#The first stage: lack of supervision and barbaric growth.The industry is rushing in the empty window of supervision, and bomb replacement products represented by JUUL are in the market.nicotine saltWith the support of technology and various seasonings, it has become extremely attractive. It has been combined with high-level marketing and extensive market cultivation, and has become popular among American teenagers. Under the catalysis of many parties, bullet-replacement products represented by mint and various fruit flavors have experienced a surge of growth, contributing most of the market increase.
thePhase 2: Implementation of supervision and structural adjustment.From a posteriori perspective, the ban on flavored and replaceable e-cigarettes was officially introduced in February 2020, and market shipment data began to reflect policy expectations at the end of 2018. The main reason is that the popularity of e-cigarettes among young people has attracted regulatory and public opinion attention. Under pressure from many parties, the leading brand JUUL revised its business strategy and gradually removed fruit-flavored and mint-flavored products from the shelves to drive the industry's cargo structure. Before the ban was officially introduced, the structural adjustment of the bomb-replacement product basically ended.
#p#Pagination Title #e#theThree stages: Continue the game and accelerate integration.The flavor ban is mainly aimed at mainstream ammunition replacement products in the market. Disposable products have escaped the edge of supervision and are rapidly emerging in gray areas, suppressing the growth of ammunition replacement products. At the same time,puff The brand represented by Bar intends to use synthetic nicotine to further circumvent regulation. On the other hand, PMTA's review of the leading brand is expected to be completely completed by the end of 2023. Altria will withdraw from JUUL's equity investment and wholly acquire NJOY, and the market will enter a new round of competition cycle.It is worth noting that behind the changes in product structure is still consumers 'choice of taste. Before flavor products are fully cleared of the market, taste still seems to be the main line driving market changes.
(2) Phase1: Lack of supervision, barbaric growth #p#pagination title #e#
flavoringScale growth led by bomb replacement products.According to CDC's four-week offline retail data (Excluding online sales and specialized marketinge-cigarette productsfog shop), the overall scale of the industry has started to grow since the end of 2017. As of October 2018, shipments in the four weeks have increased by nearly 100%. Structurally, the proportion of fruits and other flavor products increased from 22% at the beginning of 2016 to 46% in October 2018, and the proportion of mint flavor products increased from 3% to 23%.The proportion of tamper-replaceable products has increased from 56% to 85%. Amper-replaceable e-cigarettes with various flavors are the main driving factor for this round of growth.
Hazard reduction #p#pagination title #e#Driven industry switching:In June 2017, the U.S. Food and Drug Administration (FDA) stated it planned to promote a tobacco nicotine harm reduction strategy,It is intended to switch from traditional cigarette users to non-combustible tobacco products (mainly atomized e-cigarettes)Reduce the health risks to tobacco users.
usePlace restrictions are more relaxed:As of April 2022, more than half of the states in the United States have implemented smoking bans in workplaces, restaurants and bars,Fewer than half of states have the same restrictions on e-cigarettesIt is expected that the smoking ban will be more relaxed in 2016-18 than the current one, objectively promoting demand for e-cigarette products.
Blank window period for listing supervision:The United States officially launched e-cigarette regulation in May 2016, and all products marketed after February 15, 2007 must undergo PMTA (Premarket Tobacco Product Application) review. But in order to regulate and encourage the development of innovative products that may be less dangerous than cigarettesnew tobaccoTo achieve an appropriate balance between products, the PMTA submission deadline has undergone three changes, and the final deadline is September 9, 2020. For the industry from 2016 to 2018, supervision is relatively remote and is a golden period for rough development.
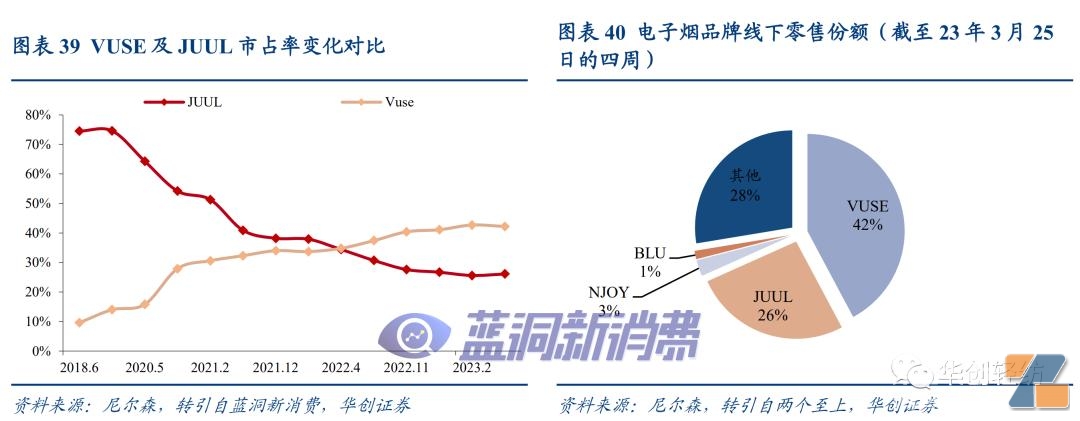
#p#Pagination Title #e#JUUL: Emerging suddenly, with a market share of up to 72%.According to sen's offline retail data,e-cigarette brandsJUUL's market share (sales) began to fluctuate and increase at the end of 2016. Growth accelerated in 2018, reaching 72% at the end of 2018. Squeezed by JUUL, the shares of other e-cigarette brands declined during the same period, JUUL became the core driver leading the first round of industry expansion.
Innovative appearance and a head start.The shape of early e-cigarette products was mainly a cylindrical shape (cigar form) that simulated traditional cigarettes, while Juul led e-cigarettes to the design trend of consumer electronics products. The appearance adopts a space aluminum body, the design adopts a U disk shape, and provides a variety of colors to choose from, which is particularly novel compared with the appearance of e-cigarette products in the same period.

#p#Pagination Title #e#
formulaUpgrading, better experience drives higher user conversions.Traditional e-cigarettes use free alkali nicotine (free-base nicotine), JUUL pioneered the use of nicotine salt (proto) composed of nicotine and weak acidnate nicotine/ nicotine salt), the added benzoic acid will make the taste smoother and reduce tingling,At the same time, nicotine concentration and transmission efficiency are higher(The transmission speed is up to 2.7 times that of free nicotine), bringing greater addiction. In addition, based on the higher taste reduction advantage of nicotine salt, many novel fruit flavors such as JUUL have improved the experience of traditional e-cigarettes and have higher user conversion rates.

preciseMarketing eye-catching advertising to achieve consumer awareness of "brand is category".# p#pagination title #e#The first time JUUL products were introduced to the market, they focused on music festivals and other activities to achieve accurate positioning of young customers. At the same time, JUUL attaches great importance to social media and has established a marketing department for Internet celebrities to promote a free and relaxed lifestyle. It has launched a large number of advertising push on new media platforms such as Twitter, Instagram, and YouTube where young people gather. It has created multiple hashtags such as #juul,#juulvapor,#switchtojuul,#vaporized, etc. Successful marketing has made JUUL popular on social media and led the "Juuling" trend among young people.
#p#Pagination Title #e#
(3) Phase2: Implementation of supervision and structural adjustment
Supervision has been gradually advanced to implementation, and industry business strategies have been revised accordingly.The FDA's supervision of the industry has gradually been upgraded from warning letters to laws and regulations. In March 2019, a draft bill was introduced to solicit public opinions. It is planned to give priority to mandatory enforcement of flavored e-cigarette products that attract minors. The formal bill will be implemented in February 2020. Implementation, priority will be given to replacement products. During this period, the overall market shipments fluctuated, and the proportion of disposable products with fruit flavors began to increase since October 2019, reflecting market operators 'early prediction of policies and corresponding business strategy revisions.

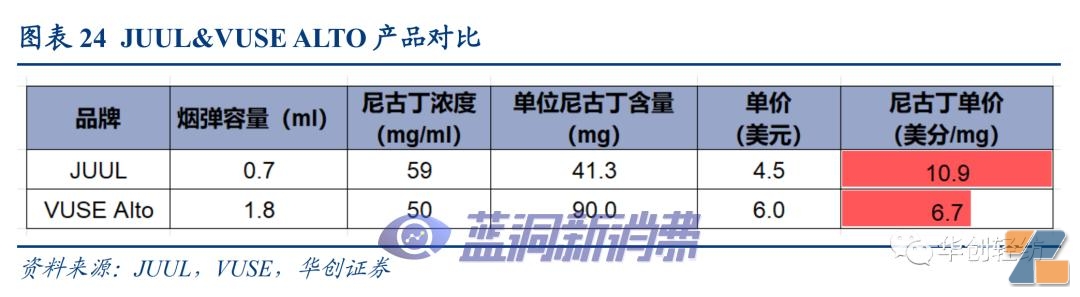
#p#Pagination Title #e#JUUL:In addition to the focus of supervision, it also faces the pressure of technological iteration.As an industry leader, JUUL has received the focus of supervision and the public. It is difficult for its product strategy to switch to fruity disposable products, because this will also lead to the risk of inducing minors to use them, and it will still face compliance pressure in the future. In addition, Reynolds Tobacco (a subsidiary of British American Tobacco)Adopt Simmore International'sFEELMVUSE Alto for ceramic core products was launched in September 2018Benefiting from the upgrade of heating technology, the product's pumping experience is better than JUUL products equipped with traditional cotton cores, and the terminal retail price is also advantageous than JUUL, and JUUL's share has begun to decline.。

#p#Pagination Title #e#
(4) Phase3: Continue the game and accelerate integration
The game of equipment types continues to suppress shipments of e-cigarettes.Since March 2020, disposable products have continued to be popular among young users with their more attractive various flavoring products. With the shipment of squeeze and replacement e-cigarettes, PUFF BAR, the disposable e-cigarette brand, has become the most popular e-cigarette product in the United States.The popularity of disposable e-cigarette products in the market is a comprehensive result of slow enforcement of regulatory loopholes. Specific reasons include:
2020The ban on flavored e-cigarettes officially introduced in February 2008 mainly targets tamper-replacement products, and disposable e-cigarettes are not explicitly included in the priority mandatory law enforcement list. At the same time, some disposable e-cigarette brands subsequently used synthetic nicotine salts to further evade supervision.
The large number of PMTA applications has dragged down the pace of FDA review and enforcement, and the expected approval completion date has been delayed many times.
FDA's internal personnel changes and organizational adjustments (Robert M. Califf will take office as the new director of the FDA in February 2022).
shareholdersTermination of commercial collaboration:In January 2021, Altria announced that starting from 2020 Q1, in addition to assisting JUUL in compliance matters related to PMTA applications, it will stop providing other commercial resources to JUUL, includingRetail channels, customer resources and supply chains。
PMTAApplication for failure: #p#pagination title #e#In June 2022, JUUL received an MDO (Marketing Ban) from the FDA regarding PMTA application. The main reason was that it did not fully provide the product's harm reduction certificate to the public.
Non-competitive agreement expires:In September 2022, Altria chose to withdraw from the non-competition clause signed with JUUL because JUUL's valuation was less than 10% of the consideration at the time of the acquisition.
Litigation pressure:Due to a large number of marketing and promotion targeted at minors in the past, JUUL has faced lawsuits from different social entities such as states and local schools. According to the JUUL Lab official website announcement on April 25, 2023,It has reached settlements with 48 states and regions, spending a total of US$1 billion and solving youth use problems.。
02
Outlook:Supply side is cleared, head replacement brands benefit from the recovery of scale and pattern is optimized
(1) OnceSex: The underlying logic of clearing is clear, and the implementation of supervision promotes share switching
The ban on flavored e-cigarettes officially introduced in February 2020 mainly targets replaceable products. Disposable products are in a regulatory gray area. Since then, some brands have further circumvented regulation by replacing nicotine extracted from tobacco with synthetic nicotine (the past U.S. tobacco policy framework only includes nicotine extracted from tobacco into the regulatory system).
 #p#Pagination Title #e#
#p#Pagination Title #e#
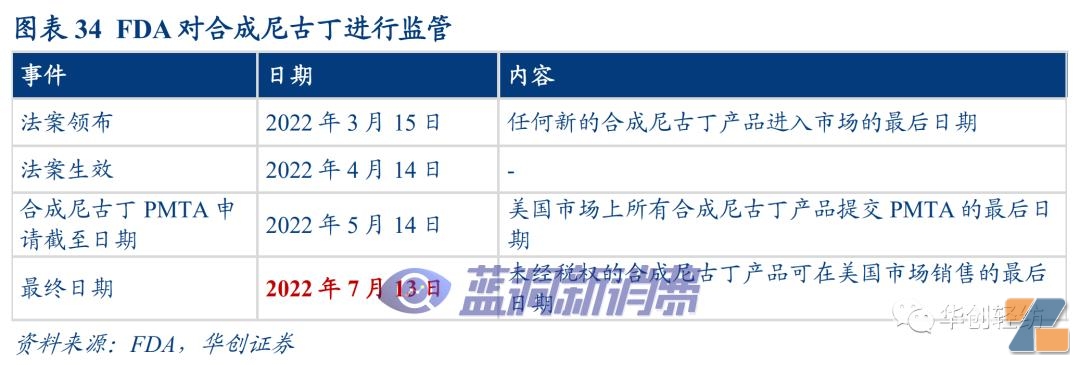
toFor example, the one-off brand PUFF BAR became popular among American teenagers in 2020 and has also received regulatory focus. In July 2020, PUFF BAR stated on its official website that it would stop all online and channel distribution. In February 2021, PUFF newly introduced e-cigarette products with synthetic nicotine, and stated that the new products "do not contain tobacco or any tobacco extracts." On March 15, 2022, the U.S. policy signed a relevant decree to include synthetic nicotine in the scope of FDA regulation and require synthetic nicotine products to submit a PMTA application before May 14, 2022. July 13, 2022 is the last date for unauthorized related products to be sold in the United States.
due toThe pace of PMTA review has dragged down the FDA's enforcement process. The statistics of single-use products from July to December 2022 have not changed significantly, but the positive changes have gradually been reflected: 1) In February 2023, Renault Tobacco submitted a citizen's petition requestingFDA issues mandatory enforcement order against disposable e-cigarettes。2) In 2023, the official website of the first e-cigarette brand PUFF BAR has listed allNicotine-containing e-cigarette products were removed from the shelves and instead sold nicotine-free "atomized" electronic products that were free of addiction.
#p#Pagination Title #e#

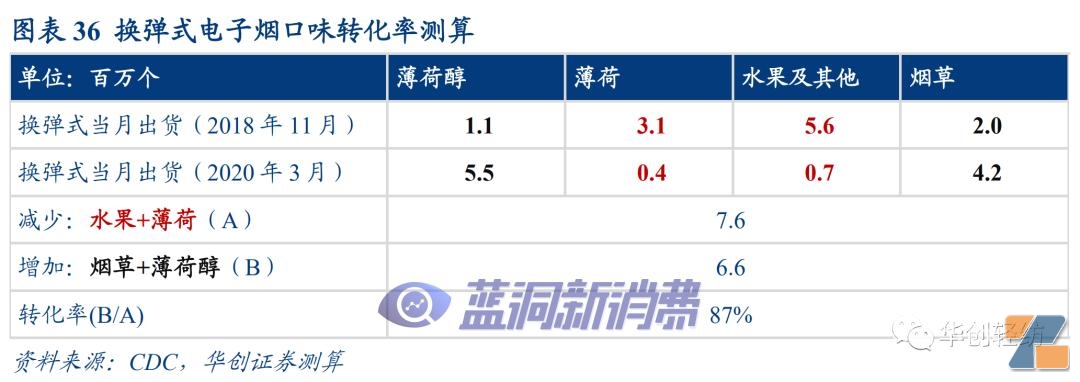
turnThere are two time nodes for calculating the transformation rate:November 2018 (before the major adjustment of the flavor of the bomb replacement style) and March 2020 (the flavor ban was implemented and the structural adjustment was completed). Market status at this time:Disposable products have not exploded on a large scale and generated significant external disturbances, and the internal users and tastes of the re-loaded e-cigarette products have once again been matched and rebalanced.
Variables that may affect the actual calculation of conversion rates:1) As the penetration rate of e-cigarettes increases and the natural growth of the industry scale, it is expected that this will increase the calculated conversion ratehigh;2) In March 2020, there has been a certain increase in flavored disposable e-cigarettes compared with November 2018, disturbing the internal conversion of tamper-replacement products, which will cause the calculated conversion rate #p#pagination title #e#low。

The original deadline for the court to require the FDA to complete the PMTA review was September 2021, which is one year after the deadline for PMTA applications. However, due to the large number of applications (as of March 31, 2023, the FDA had completed 6.7 million PMTA reviews submitted before September 9, 2020), and legal challenges (some companies chose to appeal after receiving an MDO), the approval pace lags behind.# p#pagination title #e#
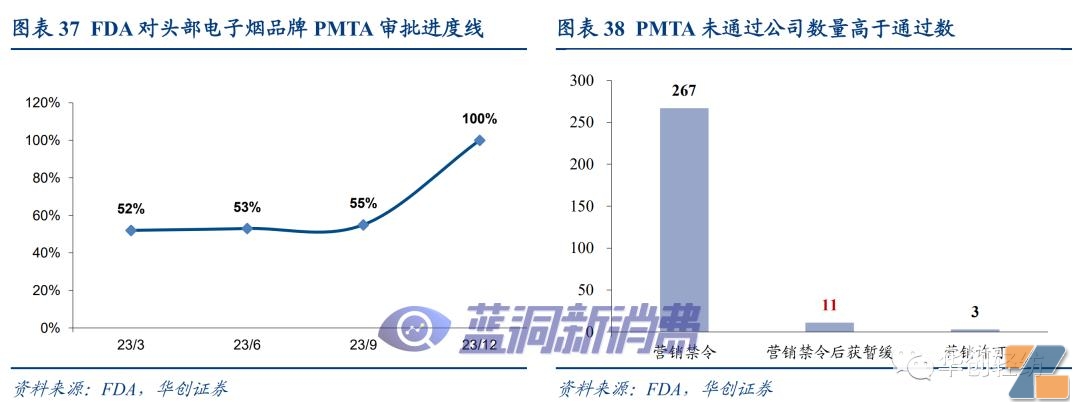
The FDA is currently promoting recommendations for head brand products (Juul, Vuse, Njoy, Logic, Blu, Smok, suorin, owned by the Puff Bar brand and accounted for more than 2% of Nielsen's offline retail data) PMTA review.As of March 31, 2023, 52% of relevant coverage applications have been completed, and it is expected that all coverage applications will be completed by the end of 2023, and the approval status report will be regularly updated in accordance with court requirements.
Judging from the existing approval results, the FDA review process is strict, especially the product's harm reduction to adult smokers and the scientific demonstration of the protection of minors. Currently, the number of companies that have obtained marketing licenses is far lower than the marketing ban. It is expected that the law enforcement of relevant unauthorized products will become the focus of supervision after the review is completed. The interior of replacement products will be gradually cleared out. VUSE, represented by the big tobacco company, focuses on compliance management Brands are expected to benefit from industry integration.

#p#Pagination Title #e#
JUUL's strategy is shrinking and NJOY is expanding soon.In September 2022, tobacco company ach and JUUL ended their non-competition agreement,In November 2022, JUUL laid off 400 people to reduce operating budget by 30%-40%In March 2023, Alachchi withdrew its minority investment in JUUL and issued an announcement in the same month.The wholly-owned acquisition of NJOY, the third brand with the market share of the cartridge replacement, has become a new platform for Altria to expand its atomized e-cigarette business.
Transaction overview:The wholly-owned acquisition of NJOY for US$2.75 billion in cash, with an additional 500 million in cash payment, based on the PMTA review of Njoy ACE menthol and other products.
NJOYOverview:NJOY ACE is currently the only ceramic core product in the United States that has passed PMTA, accounting for 85% of NJOY's shipments in 22 years. It has low penetration and strong compliance among young customers. In terms of channels, ACE has 33000 retail terminals (VUSE:121000), accounting for 3% of the U.S. retail market share (VUSE: 35%).
Acquisition considerations:1) NOJY ACE successfully passed PMTA. 2) NJOY products perform well in protecting minors. 3) Alachchi seller survey shows that NJOY has a higher acceptance among adult smokers. 4) Alachchi's commercial resources can effectively empower NJOY and accelerate its scale expansion in the adult market.
commercialCollaboration:Alacher is committed to maintaining the business cooperation relationship between NJOY and Simmore International, and will fully empower NJOY in terms of marketing experience, retail network, and sales team, focusing on NJOY's brand expansion among adult smokers in the United States.
#p#Pagination Title #e#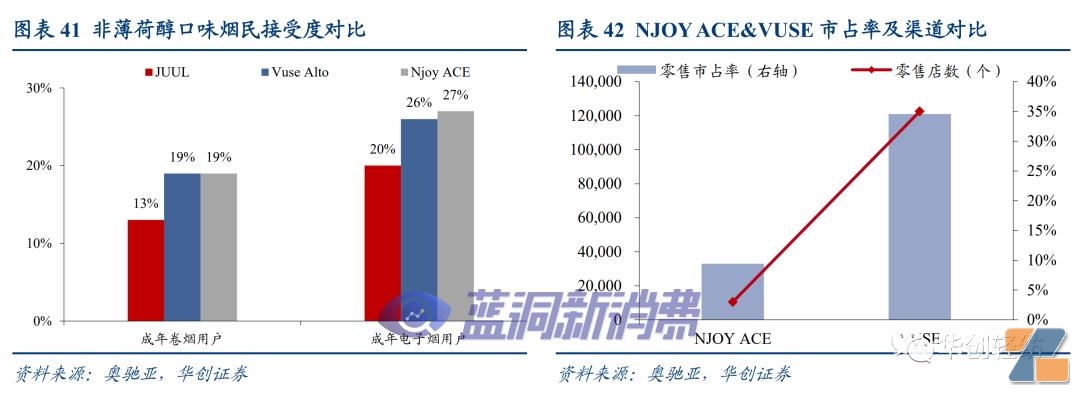
03
Simmore International:Binding head brands is expected to fully benefit from industry integration
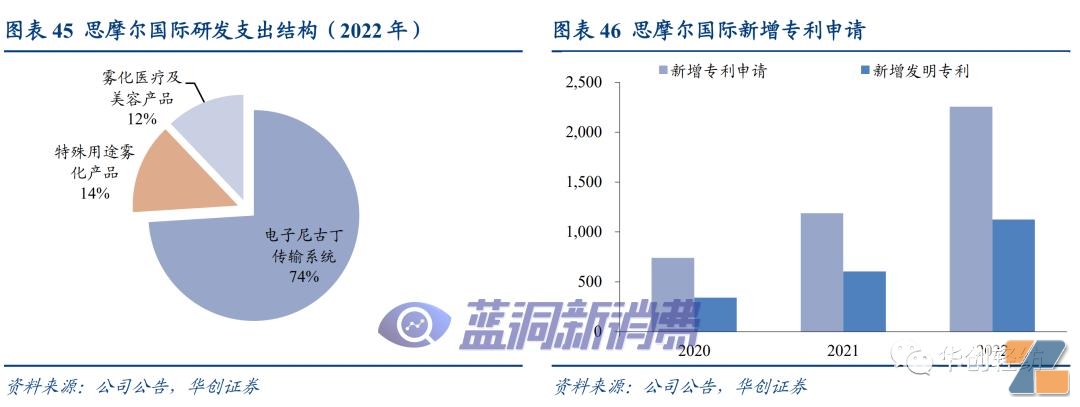
Binding head brands is expected to fully benefit from industry integration.As the core supplier of VUSE and NJOY, leading brands in the U.S. market, Simmore International continues to deepen R & D and manufacturing and strengthens cooperation and binding with major customers in compliance operations. In the past, revenue growth in the United States was disrupted by illegal operations of one-time products. In the future, it is expected to benefit from the internal integration of one-time product clearance and replacement products, share the market expansion dividends of downstream customers, and the flexibility of manufacturing share is worth looking forward to.
#p#Pagination Title #e#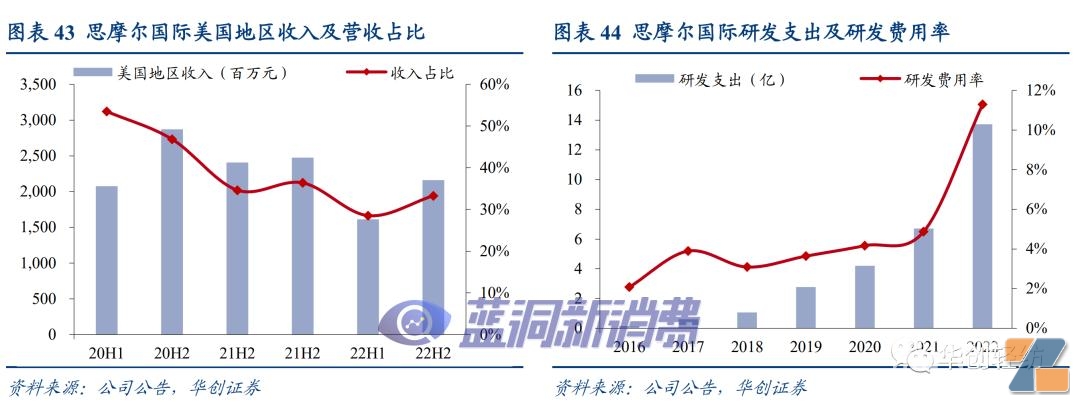
04
Risk Warning
Menthol products have not been reviewed:At present, no menthol flavor products have passed the PMTA review. If PMTA prohibits the marketing of menthol products in the future, it may affect the overall market size.



